Chemistry
Atomic number of element M is 12 and it forms an ionic compound with element L.
(a) Which of the following atomic numbers will match L?
i. 14
ii. 10
iii. 8
(b) What is the name given to the members of the group to which element M belongs?
(c) Draw the electron dot structure of the compound formed between M and L.
Answer
Atomic number of element M = 12
Electronic configuration of element M = 2, 8, 2
∴ Element M is a metal with valency 2, it will form M2+ ion
(a)
| Atomic Number | Configuration | Evaluation |
|---|---|---|
| 14 | 2, 8, 4 | Tends to share electrons and forms covalent bonds |
| 10 | 2, 8 | Noble gas – inert, does not form ions |
| 8 | 2, 6 | Non-metal, gains 2 electrons to form ionic compounds |
The correct match for L will be (iii), because M can form an ionic compound with L (M2+ + L2- = ML)
(b) As element M contains 2 valenced electrons, it belongs to Group 2 of the Periodic Table and group 2 elements are called Alkaline Earth Metal.
(c) The electron dot structure of the compound formed between M and L is shown below:
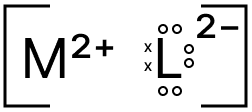
Related Questions
The empirical formula of a hydrocarbon is C2H3. The hydrocarbon has a relative molecular mass of 54. (At wt: H = 1, C = 12)
(a) What is the molecular formula of the hydrocarbon?
(b) Draw the structural formula of the hydrocarbon.
(c) Give the general formula of the hydrocarbon.
Study the given figure and answer the given questions:
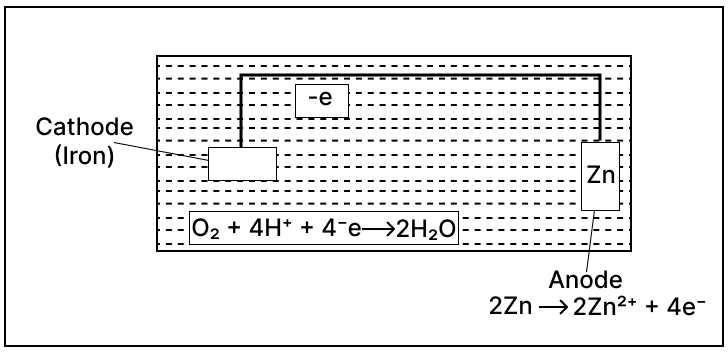
(a) Identify the application of electrolysis demonstrated above.
(b) Which metal is protected in the above process?
(c) Why should the metal be protected?
Nita's father bought a basket of ripe mangoes. While opening it she found a small sachet containing a white crystalline powder along with the mangoes. She was told that it is a chemical that releases a gas when it comes in contact with moisture, that induces ripening of fruits.
(a) Name the chemical powder in the sachet.
(b) Name the gas.
(c) Give a balanced chemical equation for the reaction that results in the evolution of this gas.
When two dry gases, oxygen and X, are passed over heated platinum, reddish-brown fumes are seen in the receiving flask, as shown in the figure.
(a) Name the gas X.
(b) Give equation(s) for the reaction(s) that resulted in the formation of brown fumes.