Physics
A certain nucleus P has a mass number 15 and atomic number 7.
(a) Find the number of neutrons.
(b) Write the symbol for the nucleus P.
(c) Write the symbols of the resulting nuclei in the following cases if :
the nucleus P loses :
- one proton
- one B-particle
- one α-particle
Express each change by a reaction.
Radioactivity
4 Likes
Answer
(a) The number of neutrons in a nucleus can be calculated using the formula:
Number of neutrons = Mass number (A) - Atomic number (Z)
Given,
Mass number (A) = 15
Atomic number (Z) = 7
So, number of neutrons = 15 - 7 = 8
(b) The nucleus P can be written as 157P.
(c)
After the loss of 1 proton, the mass number and atomic number of the nucleus P will decrease by 1.The new nucleus will be (say). The change can be written as :
After the loss of one β particle, the mass number will remain the same, but the atomic number will increase by 1. The nucleus changes to (say) as follows :
- After the loss of one α particle, the mass number decreases by 4 and the atomic number decreases by 2. The nucleus changes to (say) as follows :
Answered By
2 Likes
Related Questions
Two pendulums C and D are suspended from a wire as shown in the given figure. Pendulum C is made to oscillate by displacing it from it's mean position. It is seen that D also starts oscillating.

(i) Name the type of oscillation, C will execute.
(ii) Name the type of oscillation, D will execute.
(iii) If the length of D is made equal to C, then what difference will you notice in the oscillations of D?
(iv) What is the name of the phenomenon when the length of D is made equal to C?
(a) Students of a class perform an experiment on series and parallel combinations of two resistances R1 and R2. Some of the students plotted graph (1) and remaining plotted graph (2). Identify the correct graph and give reason.
A
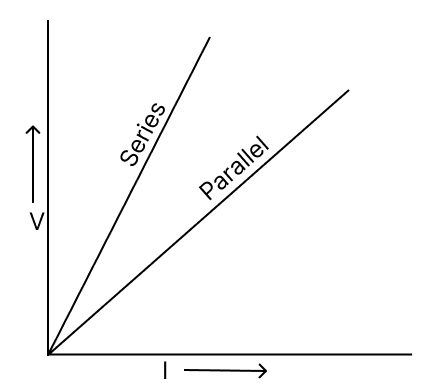
B
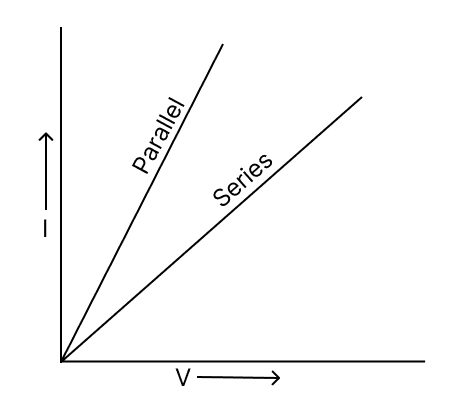
(b) The length of an electric wire is increased by 25%. What is the percentage increase in the resistance and resistivity?
(c) Same current flows through an electric live wire and a bulb filament, but only the filament glows. Give reason.
The circuit diagram shown alongside includes a 6 V battery, an ammeter A, a fixed resistor R1, of 2 Ω and resistance wire R2, connected between the terminals A and B. The resistance of the battery and ammeter may be neglected. Calculate the ammeter readings when the wire R2, is of :

(a) 0.20 m length and of resistance 4 Ω.
(b) 0.40 m length and of the same thickness and material as in case (i).
(c) 0.20 m length and having an area of cross section double than that in case (i).
Calculate the total amount of heat energy required to melt 200 g of ice at 0°C to water at 100°C.
(Specific latent heat of ice = 336 J g-1, specific heat capacity of water = 4.2 J g-1°C-1)