Chemistry
The diagram given below shows electrolysis of water. Answer the following questions :
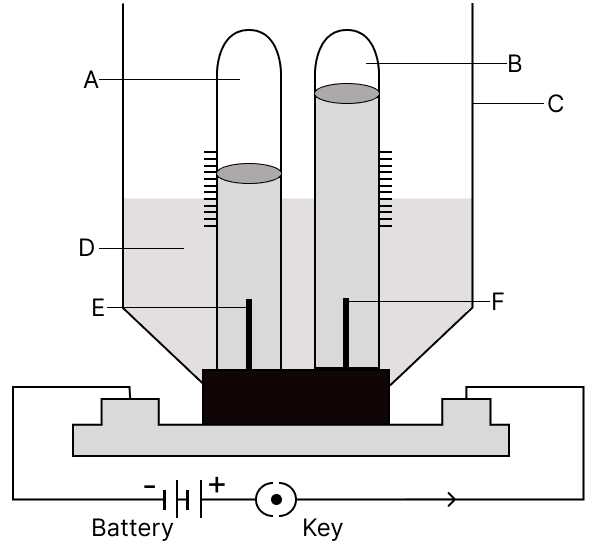
(a) Label A, B, C, D, E and F.
(b) Why is acidulated water used ?
(c) What is the ratio by volume for the gases collected ?
Hydrogen
1 Like
Answer
(a)
A → Hydrogen (H2)
B → Oxygen (O2)
C → Electrolytic Cell
D → Acidulated Water (Electrolyte)
E → Cathode
F → Anode
(b) Acidulated water is used as pure water is a bad conductor of electricity. When a small amount of acid, alkali or salt is added to it, only then it conducts electricity.
(c) Ratio by volume of the gases collected
Hydrogen : Oxygen = 2 : 1
Answered By
3 Likes
Related Questions
Give three differences between Cathode and anode.
Give three differences between Cation and anion.
(a) Name the chemicals required to prepare hydrogen gas in the laboratory.
(b) Give a balanced chemical equation for the reaction.
(c) Draw a neat and well-labelled diagram for the laboratory preparation of hydrogen.
(d) How is hydrogen gas collected? Why?
(e) Give one chemical test for hydrogen. Why?
Hydrogen is the most abundant element in the universe. On earth, however, it is rarely found in the free state because of its high reactivity, and usually occurs in combined forms such as water and hydrocarbons. Owing to its unique properties, hydrogen has a wide range of applications. It is used in cutting and welding metals, as a reducing agent for extracting metals from their oxides, in the preparation of vanaspati ghee from vegetable oils and in the manufacture of many important chemicals. Today, hydrogen is widely regarded as a potential clean fuel of the future.
(a) Why is hydrogen known as a clean fuel ?
(b) Define the process used to prepare vanaspati ghee from vegetable oils. Name a catalyst used in this process.
(c) Why is hydrogen used for cutting and welding metals ?
(d) Why is hydrogen not used in weather forecasting balloons anymore ?
(e) Name the gas which has replaced hydrogen in these balloons. Why ?