Chemistry
Distinguish the following by your observations:
(a) Calcium nitrate solution and zinc nitrate solution (using NH4OH).
(b) Sodium carbonate and sodium sulphite (using HCl).
Practical Chemistry
2 Likes
Answer
(a) When ammonium hydroxide (NH4OH) is added to zinc nitrate solution [Zn(NO3)2], a gelatinous white ppt of zinc hydroxide [Zn(OH)2] is obtained which is soluble in excess of NH4OH.
(With excess NH4OH ppt. dissolves)
On the other hand, calcium nitrate solution [Ca(NO3)2] does not give any ppt. even when excess of ammonium hydroxide is added.
(b) When dil. sulphuric acid is added to sodium carbonate and heated, colourless, odourless gas is evolved which turns lime water milky and has no effect on KMnO4 or K2Cr2O7 solutions.
Na2CO3 + 2HCl ⟶ 2NaCl + H2O + CO2 ↑
When dil. sulphuric acid is added to sodium sulphite and heated, colourless gas with suffocating odour is evolved which turns lime water milky. It turns acidified K2Cr2O7 from orange to clear green and pink coloured KMnO4 to clear colourless.
Na2SO3 + 2HCl ⟶ 2NaCl + H2O + SO2 ↑
Hence, the two compounds can be distinguished.
Answered By
1 Like
Related Questions
State your observations in each of the following:
(a) Dilute hydrochloric acid is added to calcium hydrogen carbonate.
(b) At the cathode, acidified aqueous copper sulphate solution is electrolysed with copper electrodes.
(c) Moist starch iodide paper is introduced into chlorine gas.
(d) Calcium hydroxide is heated with ammonium chloride crystals.
Draw the structure of the stable positive ion formed when sulphuric acid dissolves in water.
A cylinder contains 68 g of Ammonia gas at STP
(i) What is the volume occupied by this gas?
(ii) How many moles of ammonia are present in the cylinder?
(iii) How many molecules of ammonia are present in the cylinder?
The structures of some organic compounds are given :
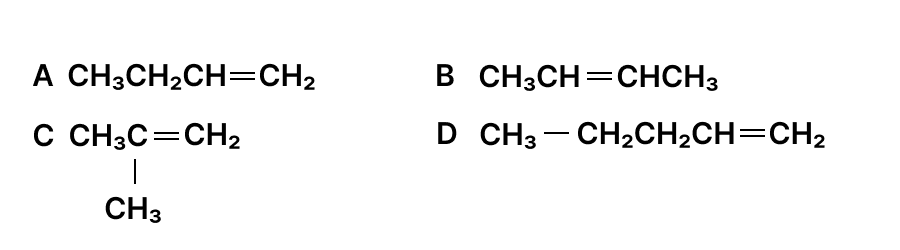
(a) Identify the isomers among the structures given above.
(b) State the members of the homologous series which are not isomers.
(c) Write the I.U.P.A.C. name of C.