Chemistry
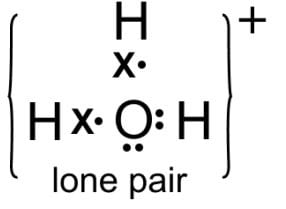
Draw the structure of the stable positive ion formed when sulphuric acid dissolves in water.
Related Questions
Write balanced equations for the following reactions:
(a) Preparation of ethyne from ethylene dibromide.
(b) Preparation of ester using carboxylic acid.
(c) Conversion of alkyl halide to ethene.
State your observations in each of the following:
(a) Dilute hydrochloric acid is added to calcium hydrogen carbonate.
(b) At the cathode, acidified aqueous copper sulphate solution is electrolysed with copper electrodes.
(c) Moist starch iodide paper is introduced into chlorine gas.
(d) Calcium hydroxide is heated with ammonium chloride crystals.
Distinguish the following by your observations:
(a) Calcium nitrate solution and zinc nitrate solution (using NH4OH).
(b) Sodium carbonate and sodium sulphite (using HCl).
A cylinder contains 68 g of Ammonia gas at STP
(i) What is the volume occupied by this gas?
(ii) How many moles of ammonia are present in the cylinder?
(iii) How many molecules of ammonia are present in the cylinder?