Science
(a) Draw two isomeric structures of Butene (C4H8)
(b) Name the following compounds :

(c) Write the chemical equations for the following reactions. Mention one essential condition each for these reactions to take place.
- Ethanol undergoes complete oxidation
- Propene undergoes hydrogenation
- Ethanoic acid reacts with ethanol
Organic Chemistry
4 Likes
Answer
(a) Butene (C4H8) has two possible structural isomers due to the position of the double bond:
- 1-butene

- 2-butene
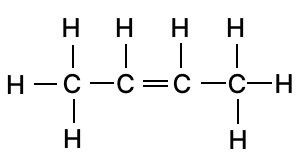
(b)
1-chloropropane
butan-2-one
(c)
1. Complete oxidation of ethanol :
Essential condition : Sufficient (excess) oxygen and ignition/heat so combustion proceeds to CO2 and H2O.
2. Hydrogenation of propene :
Essential condition : Presence of hydrogen and a hydrogenation catalyst (e.g. nickel, platinum or palladium) — often with mild heating/pressure.
3. Ethanoic acid reacts with ethanol (esterification) :
Essential condition : Acid catalyst (concentrated H2SO4) and gentle heating (reflux) to form ethyl ethanoate; removal of water drives the equilibrium to the right.
Answered By
3 Likes
Related Questions
Consider the following electric circuit :

Calculate the values of the following :
(a) The total resistance of the circuit
(b) The total current drawn from the source
(c) Potential difference across the parallel combination of 10 Ω and 15 Ω resistors
(a) Write the relationship between resistivity and resistance of a cylindrical conductor of length l and area of cross-section A. Hence derive the SI unit of resistivity.
(b) Why are alloys used in electrical heating devices?
(a) A carbon compound X is a good solvent. On reaction with sodium, X forms two products Y and Z. Z is used to convert vegetable oil into vegetable ghee. Identify and name X, Y and Z. Also write the equation of reaction of X with sodium to justify your answer.
(b) Write chemical equation to show what happens when ethanol :
- burns in oxygen/air.
- is heated at 443 K in excess conc. H2SO4.
- reacts with acidified potassium dichromate.
(a) Write the functions of the following parts of human female reproductive system :
- Ovary
- Fallopian tube
- Uterus
(b) State briefly two contraceptive methods used by human males.