Chemistry
An element X combines with oxygen to form an oxide X2O3. This oxide is a good conductor of electricity and can be reduced to its metal only by electrolysis.
(a) Write the equation for the reaction formed when the oxide (X2O3) combines with hydrochloric acid.
(b) How many valence electrons are present in the outermost shell of X?
(c) Will element X undergo oxidation or reduction?
Acids Bases Salts
1 Like
Answer
Element X is a metal, and its oxide is X2O3 so X must have a valency of 3 (X3+). The oxide is a good conductor of electricity. Since it requires electrolysis to extract X, it must be a very reactive metal.
(a) The oxide (X2O3) is reacting with hydrochloric acid, so we can say the oxide is ionic and basic in nature. The reaction will give salt and water.
X2O3 + 6HCl ⟶ 2XCl3 + 3H2O
(b) Since X forms X3+, it must lose 3 electrons, meaning it has 3 valence electrons.
(c) To form X3+, element X must lose 3 electrons and loss of electron is oxidation.
Answered By
2 Likes
Related Questions
Rohan wants to electroplate a spoon with nickel.
(a) To which electrode should he connect the article to be electroplated?
(b) Write the equation for the reaction that will occur at the cathode.
(c) What should the anode be made up of?
Amit found that 30g of a gas occupied 1000 c.c at STP.
(a) What will the gram molecular weight and the vapour density of this gas be?
(b) How many molecules of this gas will be present in 44.8 l of it?
Observe the reactions given below and answer the following questions:
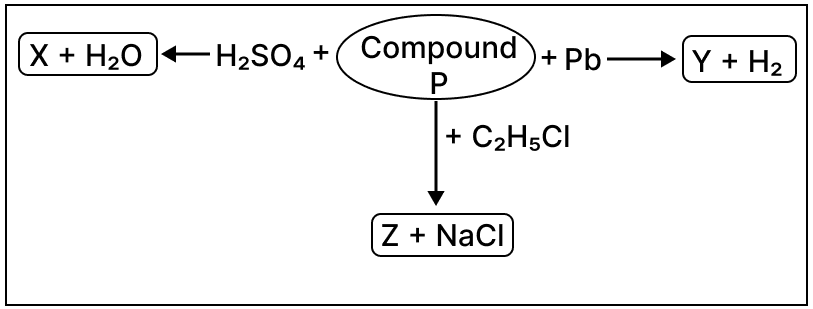
(a) Identify compound P.
(b) Give the chemical formula of Z.
(c) Write the reaction taking place between the identified compound P and sulphuric acid.
(d) Name compound Y.
Study the information given in the table below and answer the questions that follow. (Note- the letters do not represent the actual symbols of the elements)
Element Electronic configuration Ionisation energy kjmol-1 X 2,2 900 Y 2,8,2 738 Z 2,8,8,2 590 (a) Explain why element X has highest ionisation energy.
(b) To which period does Z belong?
(c) Draw the electron dot structure of the compound formed between Z and oxygen.