Chemistry
Following is a list of metals :
Na, Zn, Fe, Ag, Pb, Hg, Cu.
Answer the questions that follow :
(a) Which of the above metals can displace all the metals from their salt solution ?
(b) Name one of the above metals which cannot react with dilute acid to produce hydrogen gas.
(c) Name one of the above metals which reacts with oxygen upon heating to produce an amphoteric oxide.
(d) Arrange the above metals in a decreasing order of their reactivity.
Chemical Reaction
2 Likes
Answer
(a) Na, that is Sodium can displace other metals from their salt solutions because it is the most reactive.
(b) Silver (Ag) is very low in the reactivity series, so it does not displace hydrogen from dilute acids.
(c) Zinc (Zn) reacts with oxygen upon heating to produce an amphoteric oxide.
(d) The given metals in a decreasing order of their reactivity are:
Na > Zn > Fe > Pb > Cu > Hg > Ag
Answered By
2 Likes
Related Questions
How will you obtain?
(a) Magnesium oxide from magnesium.
(b) Silver chloride from silver nitrate.
(c) Nitrogen dioxide from lead nitrate.
(d) Zinc chloride from zinc.
(e) Ammonia from nitrogen.
Also give balanced equations for the reactions.
(a) What is meant by metal activity series?
(b) A metal P was placed in a solution of silver nitrate. After sometime, silver was deposited on metal P and the solution turned blue.
(i) Which is more reactive - metal P or silver?
(ii) What kind of reaction is this?
(iii) Give an example of this type of reaction.
Given below is a diagram showing a type of reaction between two compounds. Answer the questions that follow :
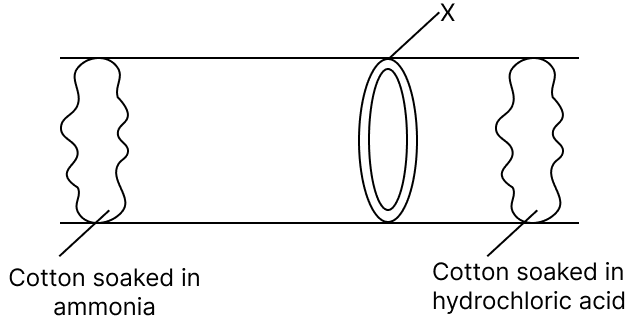
(a) What compound is X ?
(b) Give a balanced chemical equation to form X.
(c) Name the type of reaction shown.
A teacher showed a chemistry experiment in the class to her students. She poured about 100 ml of sodium hydroxide into a beaker and added a few drops of phenolphthalein to it. The colour of the solution immediately changed. Then she added dilute sulphuric acid to the beaker slowly, while constantly stirring the solution with a glass rod. She kept on adding dilute sulphuric acid until the solution became colourless again. Then she called her students to touch the beaker.
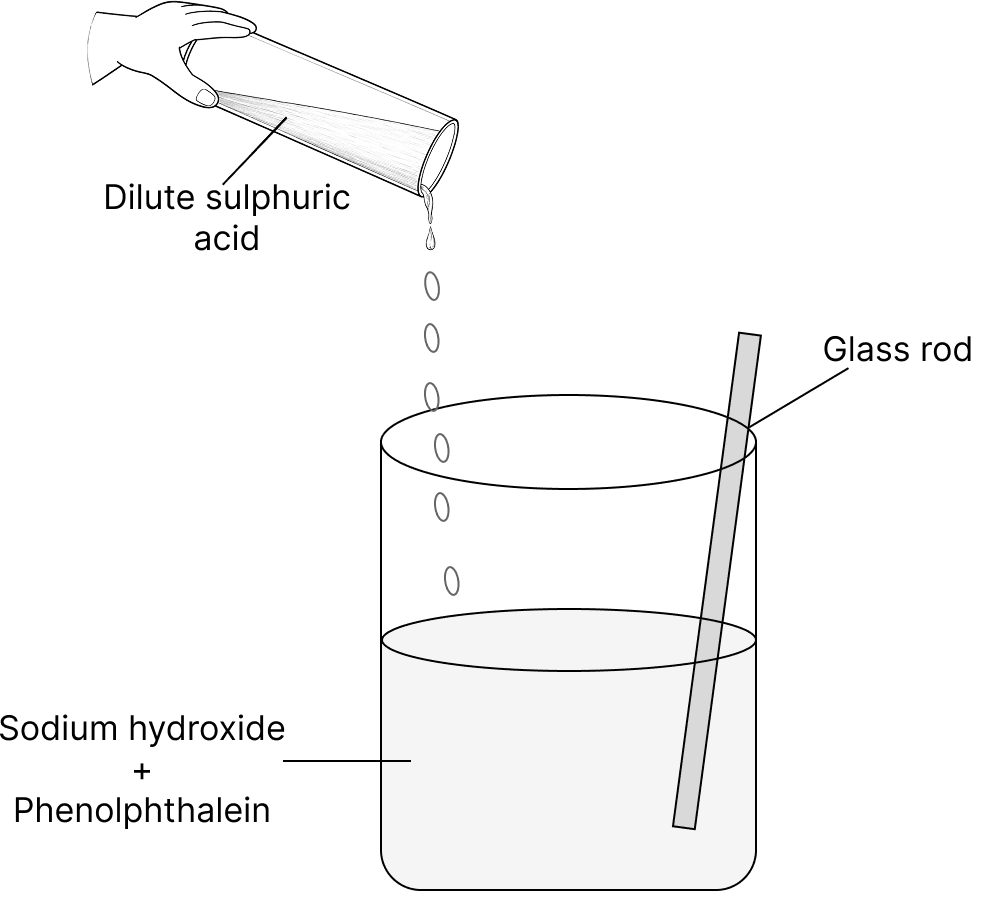
(a) Name the type of reaction which involves an acid and a base as shown by the teacher.
(b) What colour change was observed when phenolphthalein was added to the beaker?
(c) Write a balanced chemical equation for the reaction between sodium hydroxide and dilute sulphuric acid.
(d) What will the students feel on touching the beaker after the reaction? Why?
(e) Why do many toothpastes contain bases?
(f) Name:
(i) a basic substance to treat an acidic soil.
(ii) an acidic substance to treat a basic soil.