Chemistry
Give one significant observation when:
(a) Excess of chlorine gas reacts with ammonia.
(b) Zinc nitrate is strongly heated in a test tube.
Ammonia
28 Likes
Answer
(a) Colourless ammonia gas reacts with greenish yellow excess chlorine giving a yellow explosive liquid (Nitrogen trichloride).
NH3 + 3Cl2 [excess] ⟶ 3HCl + NCl3
(b) Reddish brown nitrogen dioxide gas is evolved on heating zinc nitrate crystals.
Answered By
20 Likes
Related Questions
Match the following Column A with Column B.
Column A Column B (a) Aluminium 1. Covalent compound (b) Sulphuric acid 2. Carbonate ore (c) Calcination 3. Hall Heroult's process (d) Calcium Chloride 4. Contact Process (e) Carbon tetrachloride 5. Electrovalent compound (a) Give the IUPAC name of the following organic compounds:
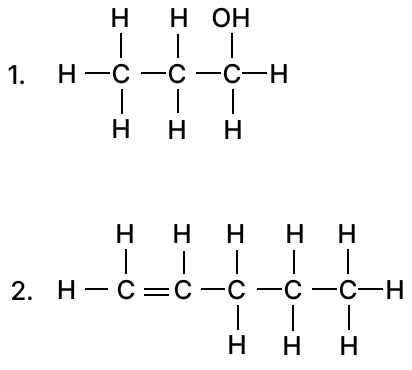
(b) Draw the structural diagram for the following compounds:
- but-2-yne
- 1, 1, 1, trichloro methane
- pentan-2-ol
Give reasons:
(a) When ammonia gas is passed over black copper oxide in a combustion tube a reddish-brown substance is left behind.
(b) Quick lime is not used to dry hydrogen chloride gas.
The electron affinity of an element X is greater than that of element Y.
(a) How is the oxidising power of X likely to compare with that of Y?
(b) How is the electronegativity of X likely to compare with that of Y?
(c) State whether X is likely to be placed to the left or to the right of Y in the periodic table?