Chemistry
Answer
| Cation | Anion |
|---|---|
| Cations are positively charged atoms or group of atoms. | Anions are negatively charged atoms or group of atoms. |
| During electrolysis, cations migrate towards the cathode. | During electrolysis, anions migrate towards the anode. |
| Examples : Na+, Ba2+, Ca2+, etc. | Examples : Cl-, OH-, Br-, etc. |
Related Questions
State four uses of hydrogen.
Give three differences between Cathode and anode.
(a) Name the chemicals required to prepare hydrogen gas in the laboratory.
(b) Give a balanced chemical equation for the reaction.
(c) Draw a neat and well-labelled diagram for the laboratory preparation of hydrogen.
(d) How is hydrogen gas collected? Why?
(e) Give one chemical test for hydrogen. Why?
The diagram given below shows electrolysis of water. Answer the following questions :
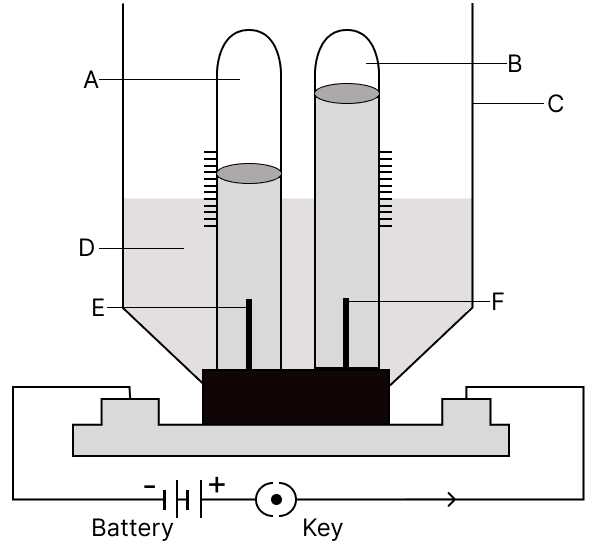
(a) Label A, B, C, D, E and F.
(b) Why is acidulated water used ?
(c) What is the ratio by volume for the gases collected ?