Chemistry
A group of students took an old shoe box and covered it with a black paper from all sides. They fixed a source of light (a torch) at one end of the box by making a hole in it and made another hole on the other side to view the light. They placed a milk sample contained in a beaker/tumbler in the box as shown in the figure. They were amazed to see that milk taken in the tumbler was illuminated. They tried the same activity by taking a salt solution but found that light simply passed through it.
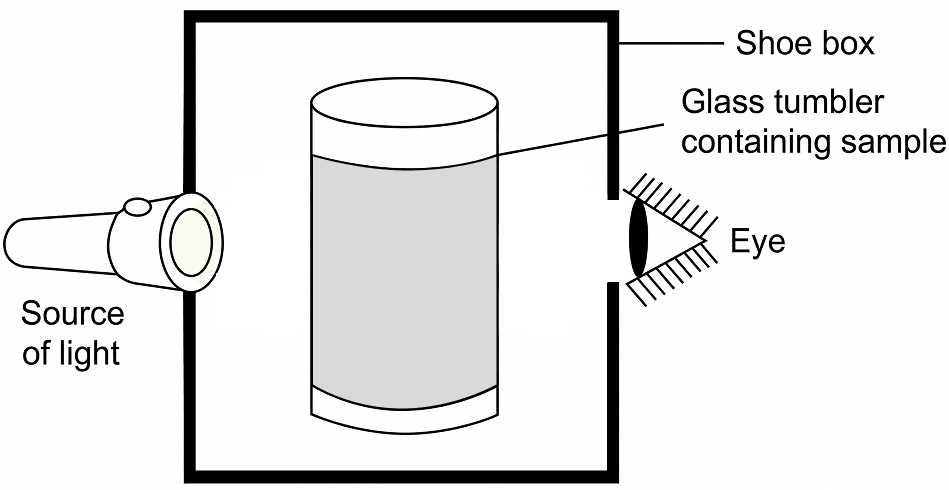
(a) Explain why the milk sample was illuminated. Name the phenomenon involved.
(b) Same results were not observed with a salt why ?
(c) Can you suggest two more solutions which would show the same effect as shown by the milk solution ?
Answer
(a) Milk is a colloidal solution. The particles of a colloid are large enough to scatter light. Therefore, when torch light passes through milk, the light gets scattered in different directions, making the path of light visible and the milk appear illuminated. This phenomenon is called the Tyndall effect.
(b) The same result is not observed with salt solution because salt solution is a true solution. Its solute particles are extremely small and do not scatter light. Hence, light passes through it without making its path visible.
(c) Two more solutions which would show the same effect are:
- Starch solution
- Soap solution
Related Questions
A solution of a solid in a liquid (such as water) can be prepared by adding the solid to water with constant stirring at a particular temperature. If the addition process is continued, a stage is ultimately reached in the dissolution process when no more of the solid dissolves. The solution at this stage is said to be saturated. The solubility of a solute is always expressed with respect to the saturated solution. It may be defined as the maximum amount of the solute that can be dissolved in 100 g of the solvent to form a saturated solution at a given temperature.
The solution becomes unsaturated if the temperature is increased. In case the temperature is decreased, the solution becomes supersaturated and crust of the solute gets deposited on the surface.(a) A student studied the solubility of three substances in water at different temperatures and collected the following data.
Temperature (°C) 286 300 320 350 Substance dissolved Solubility X 20 24 26 30 Y 30 36 48 54 Z 24 20 18 14 The correct order of the solubilities of these substances in water is :
(i) Y > X > Z
(ii) X > Y > Z
(iii) Z > X > Y
(iv) Y > Z > X(b) 10 g of a solute is dissolved in 250 g of a solvent. The solubility of the solute is:
(i) 4 g
(ii) 40 g
(iii) 10 g
(iv) 20 g(c) A saturated solution becomes unsaturated by :
(i) Heating the solution
(ii) Cooling the solution
(iii) Adding more of the solute
(iv) Adding more of the solvent(a) (i) and (iii)
(b) (ii) and (iii)
(c) (i) and (iv)
(d) (iii) and (iv)(d) Solubility of a solute in a solvent may be defined as:
(i) Dissolution of 1 g of solute in one kg of solvent at a given temperature.
(ii) Dissolution of 1 g of solute in 100 g of the solvent at a given temperature.
(iii) Dissolution of 1 g of solute in 100 mL of the solvent at a given temperature.
(iv) The maximum amount of solute which can be dissolved in 100 g of the solvent to form a saturated solution at a given temperature.Water acts as a universal solvent because it has a unique property to dissolve various organic and inorganic compounds. Its dielectric constant is high, which is about 80 at 20°C. All this is due to its polar nature.
(a) What properties of water make it an exceptional solvent compared to other solvents like ethanol, carbon tetrachloride, benzene, etc.
(b) Explain the environmental consequences of water's "universal solvent" property, particularly regarding pollution.
Name some physical and chemical processes used to make river water potable.
Which steps are most effective at removing solid impurities from water ? Explain the role of alum.