Physics
Heat energy supplied during the melting of a substance is utilised in :
- increasing the kinetic energy of molecules
- decreasing the potential energy of molecules
- increasing the potential energy of molecules
- decreasing the kinetic energy of molecules
Calorimetry
15 Likes
Answer
increasing the potential energy of molecules
Reason — When a substance is melting, it transitions from a solid phase to a liquid phase. During this phase transition, the heat energy supplied is used to overcome the forces holding the molecules together in the solid phase. This process increases the potential energy of the molecules, allowing them to break free from their fixed positions in the crystal lattice. However, the kinetic energy of the molecules remains relatively constant during the phase transition, as the temperature remains constant until the phase change is complete. Therefore, the primary use of heat energy during melting is to increase the potential energy of the molecules
Answered By
5 Likes
Related Questions
1.0 kg of water is contained in a 1.25 kW kettle. Calculate the time taken for the temperature of water to rise from 25° C to it's boiling point 100° C. Specific heat capacity of water = 4.2 J g-1 K-1.
Heat energy is …………… during melting and it is …………… during freezing at a constant temperature.
- rejected, absorbed
- rejected, rejected
- absorbed, absorbed
- absorbed, rejected
The curve which shows the rise of temperature with the amount of heat supplied for a piece of ice is :
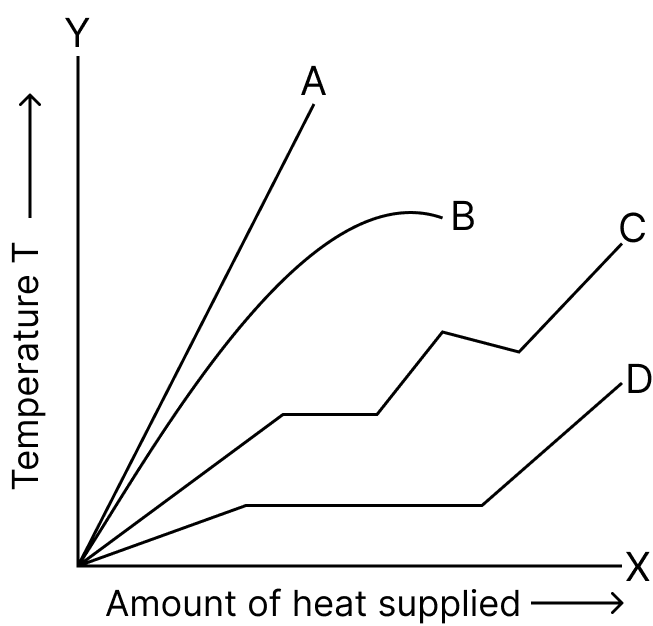
- A
- B
- C
- D
Two bodies A and B are of the same material but A has twice the mass of B. Both are heated up to the same temperature. Identify the correct statement ?
- Heat absorbed by A is half of that by B.
- Heat absorbed by A is equal to that by B.
- Heat absorbed by A is twice of that by B.
- Heat absorbed by A is four times that by B.