Chemistry
How would you show that hydrogen:
(a) is a non-supporter of combustion?
(b) is lighter than air?
Hydrogen
26 Likes
Answer
(a) Hydrogen gas is a non-supporter of combustion is shown by the following experiment:

- Hold a hydrogen gas filled jar with its mouth downwards.
- Placed a lighted candle and cover it with this jar.
- The candle gets extinguished but the gas burns with a pop sound.
This shows that hydrogen gas is non-supporter of combustion but itself is combustible.
(b) Hydrogen gas is lighter than air is shown by the following experiment:

- Take a delivery tube and place one of its ends in a liquid soap solution kept in a trough and other end inside a flat bottom jar.
- In the flat bottom jar hydrogen gas is freshly prepared by the action of dilute hydrochloric acid on zinc granules.
- Hydrogen gas enters the soap solution through delivery tube.
- The soap bubbles containing hydrogen rise upward in the air.
The rising soap bubbles prove that hydrogen is lighter than air.
Answered By
14 Likes
Related Questions
Give three differences between Cathode and anode.
Give three differences between Cation and anion.
(a) Name the chemicals required to prepare hydrogen gas in the laboratory.
(b) Give a balanced chemical equation for the reaction.
(c) Draw a neat and well-labelled diagram for the laboratory preparation of hydrogen.
(d) How is hydrogen gas collected? Why?
(e) Give one chemical test for hydrogen. Why?
The diagram given below shows electrolysis of water. Answer the following questions :
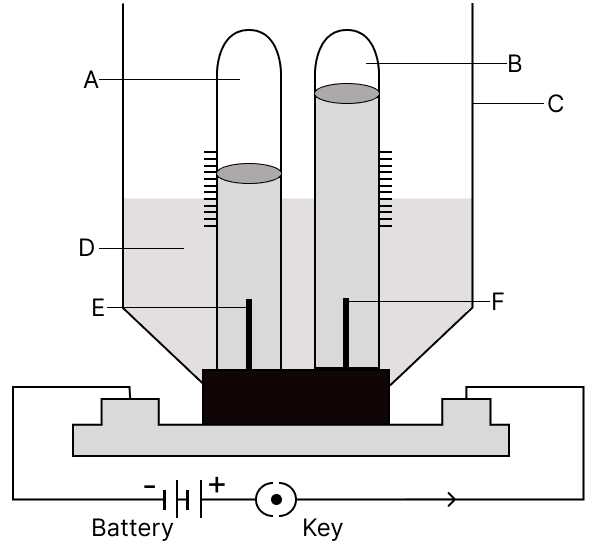
(a) Label A, B, C, D, E and F.
(b) Why is acidulated water used ?
(c) What is the ratio by volume for the gases collected ?