Chemistry
(a) What is meant by metal activity series?
(b) A metal P was placed in a solution of silver nitrate. After sometime, silver was deposited on metal P and the solution turned blue.
(i) Which is more reactive - metal P or silver?
(ii) What kind of reaction is this?
(iii) Give an example of this type of reaction.
Answer
(a) A list in which the metals are arranged in a decreasing order of their chemical reactivity is called the metal activity series.
(b)(i) Metal P is more reactive as it displaced silver from the solution of silver nitrate, to form new compound.
(ii) In this reaction, metal P displaces silver from its salt (silver nitrate, AgNO₃) to form metal P nitrate and silver metal. Hence, the given reaction is a Displacement reaction.
(iii) Zinc being more reactive than copper, displaces copper from copper sulphate solution. The blue copper sulphate solution turns colourless due to the formation of zinc sulphate and a reddish brown copper is deposited on zinc.
Zn + CuSO4 (aq) → ZnSO4 (aq) + Cu
Related Questions
Define exothermic and endothermic reactions. Give two examples of each.
How will you obtain?
(a) Magnesium oxide from magnesium.
(b) Silver chloride from silver nitrate.
(c) Nitrogen dioxide from lead nitrate.
(d) Zinc chloride from zinc.
(e) Ammonia from nitrogen.
Also give balanced equations for the reactions.
Following is a list of metals :
Na, Zn, Fe, Ag, Pb, Hg, Cu.
Answer the questions that follow :
(a) Which of the above metals can displace all the metals from their salt solution ?
(b) Name one of the above metals which cannot react with dilute acid to produce hydrogen gas.
(c) Name one of the above metals which reacts with oxygen upon heating to produce an amphoteric oxide.
(d) Arrange the above metals in a decreasing order of their reactivity.
Given below is a diagram showing a type of reaction between two compounds. Answer the questions that follow :
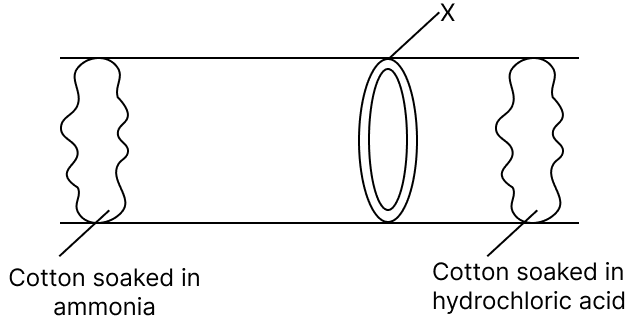
(a) What compound is X ?
(b) Give a balanced chemical equation to form X.
(c) Name the type of reaction shown.