Science
The metals obtained from their molten chlorides by the process of electrolytic reduction are :
options
- Gold and silver
- Calcium and magnesium
- Aluminium and silver
- Sodium and iron
Metallurgy
1 Like
Answer
Calcium and magnesium
Reason — Metals like calcium and magnesium are highly reactive and are obtained by the electrolytic reduction of their molten chlorides (CaCl2 and MgCl2).
During electrolysis, the metal ions are reduced at the cathode to form the pure metal and less reactive metals like gold, silver or iron are extracted by other methods and not by electrolysis of molten salts.
Hence, the metals obtained from their molten chlorides by the process of electrolytic reduction are calcium and magnesium.
Answered By
1 Like
Related Questions
Consider the following reactions :
(a) Dilute hydrochloric acid reacts with sodium hydroxide.
(b) Magnesium oxide reacts with dilute hydrochloric acid.
(c) Carbon dioxide reacts with sodium hydroxide.
It is found that in each case :
options
- Salt and water is formed.
- Neutral salts are formed.
- Hydrogen gas is formed.
- Acidic salts are formed.
Reaction between two elements A and B, forms a compound C. A loses electrons and B gains electrons. Which one of the following properties will not be shown by compound C?
options
- It has high melting point.
- It is highly soluble in water.
- It has weak electrostatic forces of attraction between its oppositely charged ions.
- It conducts electricity in its molten state or aqueous solution
The formation of magnesium oxide is correctly shown in option :
options
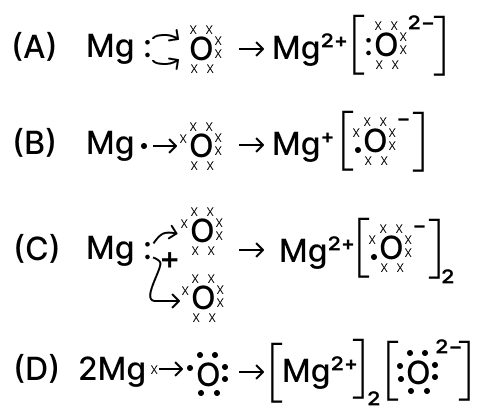
Secretion of less saliva in mouth will effect the conversion of :
options
- Proteins into amino acids
- Fats into fatty acids and glycerol
- Starch into simple sugars
- Sugars into alcohol