Chemistry
Name
(a) a carbonate which does not decompose on heating.
(b) a nitrate which produces oxygen as the only gas.
(c) a compound which produces carbon dioxide on heating
(d) a nitrate which produces brown gas on heating.
Chemical Reaction
132 Likes
Answer
(a) Sodium carbonate
(b) Potassium nitrate
(c) Calcium carbonate
(d) Lead nitrate
Answered By
71 Likes
Related Questions
The figure given below demonstrates the preparation of oxygen.
2KClO3 (s) 2KCl (s) + 3O2 (g)
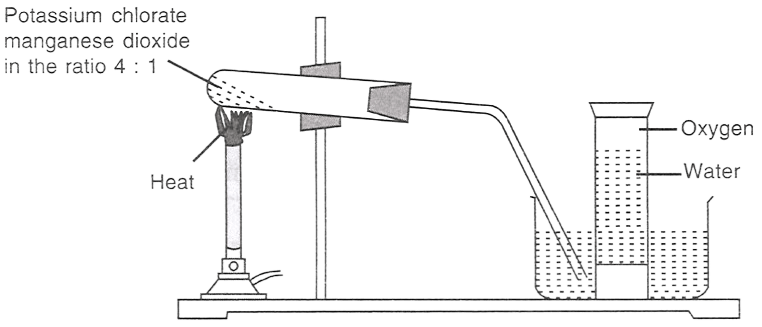
(I) Which of the following statements is/are correct about the reaction?
It is a decomposition reaction and is exothermic.
It is a decomposition reaction and is endothermic.
It is a photochemical decomposition reaction.
It is a combination reaction.
(II) Potassium chlorate is:
an oxidising agent.
a reducing agent.
both reducing as well as an oxidising agent.
all options are correct.
(III) In the reaction, manganese dioxide:
takes part in the reaction.
acts as a catalyst.
decreases the speed of reaction.
helps in producing more oxygen.
What happens when a solution of an acid is mixed with a solution of a base in a test tube?
P — The temperature of the solution increases.
Q — The temperature of the solution decreases.
R — The temperature of the solution remains the same.
S — Salt formation takes place.
Only P
P and R
Q and R
P and S
Which is the most reactive element in this question :
CuSO4 + Fe ⟶ FeSO4 + Cu
Define :
(a) Photochemical reaction
(b) Electrochemical reaction
Give one example in each case.