Chemistry
Some salts while crystallising out from their solutions, combine with a definite quantity of water. Some of these salts lose this water on exposure to dry air. Certain water soluble substances, when exposed to atmosphere at ordinary temperatures, absorb moisture from atmospheric air forming a saturated solution.
(a) State the term used for a 'definite quantity of water' combined with a salt.
(b) What type of salts lose their water molecules ? Give two examples.
(c) Table salt forms a solution on exposure to air during rainy season. Explain.
(d) How can pure sodium chloride be obtained from the above table salt solution in (c) ?
Water
3 Likes
Answer
(a) The definite quantity of water combined with a salt is called Water of Crystallisation.
(b) Salts that lose their water molecules on exposure to air are called Efflorescent salts.
Washing soda and magnesium sulphate heptahydrate are the two examples.
(c) Table salt forms a solution in the rainy season because it contains impurities like Magnesium chloride, which is deliquescent. It absorbs moisture from the air and dissolves in it, forming a solution.
(d) Pure sodium chloride can be obtained by evaporation or crystallisation.
Answered By
1 Like
Related Questions
Explain, with equations, why ordinary soap does not lather easily in hard water.
Explain:
(a) The use of lead pipes for drinking water supply is being discontinued.
(b) Chalk hills dissolve in rain water.
(c) Hard water is unfit for boilers.
(d) Iron chloride forms a saturated solution when exposed to the atmosphere.
(e) A bottle containing concentrated H2SO4 should be stoppered.
The following figure shows the solubility curves of NaCl, KNO3 and hydrated calcium sulphate.
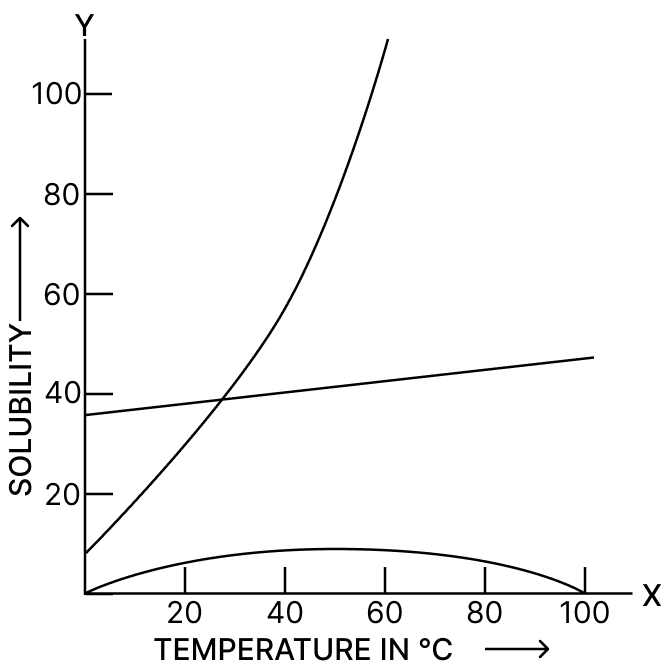
(a) Identify and label the curves with the salt it represents.
(b) State the factors on which the solubility depends.
(c) Solubility of which salt(s) shows :
(i) Endothermic process
(ii) Exothermic process?
Assertion (A): Water is a universal solvent.
Reason (R): Water dissolves all substances except noble metals and glass.
- Both A and R are true and R is the correct explanation of A.
- Both A and R are true but R is not the correct explanation of A.
- A is true but R is false.
- A is false but R is true.