Physics
A science experiment was conducted by a man in his kitchen. He kept some ice cubes at room temperature. Ice cubes melted into water. Then he placed this water in a pot on the stove and turned on the heat. As the water started to heat up, he observed some changes. As the temperature increased, he observed that the water started to form tiny bubbles and steam began to rise. Then, he placed a metal lid on top of the pot to cool down the steam. After a few minutes, when he removed the lid, he saw water droplets on the inner side of the lid.
He plotted the following graph. Observe the graph carefully and answer the following questions.
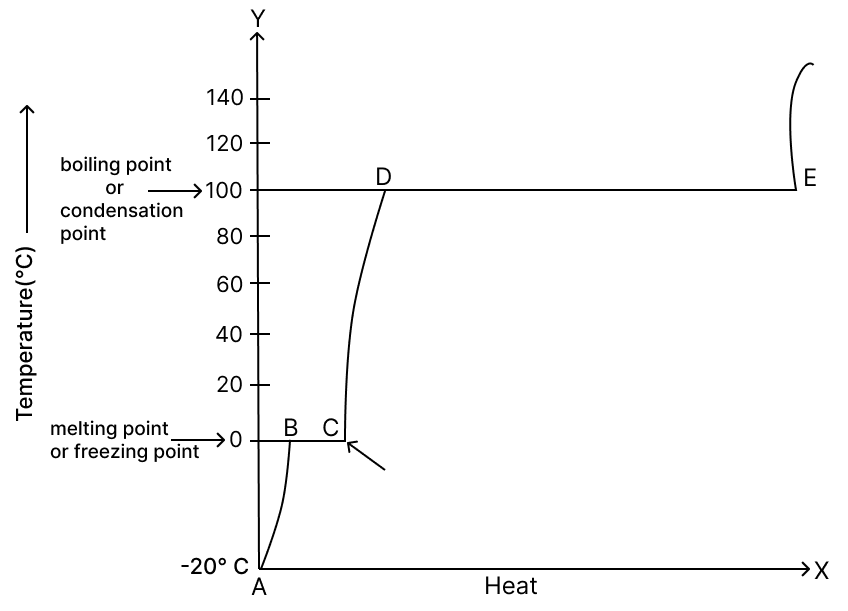
(a) What experiment did the man perform?
(b) Label A, B, C, D and E in the graph.
(c) Why did he observe water droplets on the lid?
(d) At what temperature does water change into ice?
(e) Why does it take so long to warm water in winter?
(f) Does the quantity of water also change with its state?
Answer
(a) The man performed an experiment to show the changes of state of water — solid (ice) → liquid (water) → gas (steam) → liquid again (condensation).
(b) Likely graph labels:
- A – Ice (solid) being heated (temperature rising from below - 20°C to 0°C)
- B – Melting point (0°C) – ice starts melting
- C – Water (liquid) being heated (temperature rising from 0°C to 100°C)
- D – Boiling point (100°C) – water starts boiling and turning to steam
- E – Steam (gas) being heated further above 100°C
(c) He observed water droplets on the lid because steam cooled down on touching the cold metal lid, changing from gas to liquid. This process is called condensation.
(d) Water changes into ice at 0°C (freezing point).
(e) It takes longer to warm water in winter because in winter, the starting temperature is lower so heat require to warm the water will be greater.
(f) No, the quantity (mass) of water does not change when it changes state. Only the arrangement and movement of molecules change.
Related Questions
We can move our hands through air and water, but not through a wooden structure. Why?
In which of the following cases will the sugar dissolve in a glass of water faster, and why?
Case 1: Water in the glass is cold
Case 2: Water in the glass is warmWhat happens to the volume of a substance when it changes from a solid to a liquid?
Can ice be changed directly into water vapour?