Chemistry
State the important information obtained from the following in four points:
(a) A molecular formula of a compound.
(b) A balanced chemical equation for a reaction.
Chemical Equation
3 Likes
Answer
(a) The molecular formula of a compound gives the following information:
- It represents one molecule of a compound.
- The elements or ions present in the compound.
- The number of each kind of atoms and ions present in one molecule of a compound and their ratio.
- The molecular formula enables us to calculate the mass of one molecule of the compound if the atomic mass of each atom present in the molecule is known. Molecular mass is the algebraic sum of the masses of all the atoms present in a given molecule.
(b) A balanced chemical equation gives the following information:
- It shows which substances are taking part in a chemical reaction and what products are obtained as a result of it (Qualitative).
- It shows both the number of molecules and the number of atoms of each type involved in the reaction.
- It enables us to calculate the actual amount (mass) of reactants and products involved and formed in the reaction if atomic mass of each of the elements involved in the reaction is known (Quantitative).
- It makes the study of chemistry universally standardized.
Answered By
1 Like
Related Questions
What is variable valency? Give two examples of elements showing variable valency.
(a) What is a chemical equation?
(b) Why is it necessary to balance a chemical equation?
(c) What are the limitations of a chemical equation?
State five ways by which a chemical equation can be made more informative.
Given below are two groups showing basic and acid radicals. Based on these two groups, answer the questions that follow :
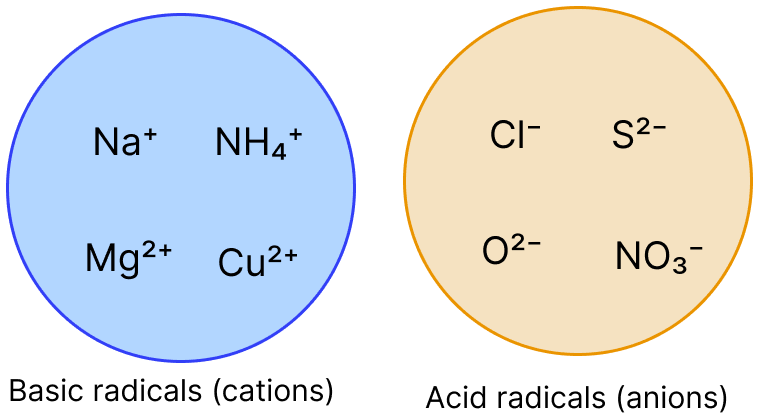
(a) Which basic radical or cation exhibits variable valency ?
(b) How is NH4+ ion different from other basic radicals in the group ?
(c) Which acid radical or anion is the odd one out in the group ? Justify your answer.
(d) Using one radical from each group, form a compound that is commonly used in daily life. Write its name and chemical formula.