Physics
The two metals A and B have their specific heat capacities in the ratio 2:3. If they are supplied the same amount of heat, then
(a) which metal piece will show a greater rise in temperature if their masses are the same?
(b) calculate the ratio in which their temperatures rise, if the mass ratio of metal A and metal B is 3:5.
Calorimetry
21 Likes
Answer
The rise in temperature is given by: ΔT =
Where:
- Q = heat supplied (same for both)
- m = mass
- c = specific heat capacity
- Δ T = temperature change
(a) Given that masses are same,
Since:
here Q and m are the same for both metals A and B, so
So,
Metal A shows a greater temperature rise than metal B (since it has lower specific heat).
(b) Masses in ratio 3 : 5 Now :
The ratio of temperature rise is 5 : 2
Answered By
12 Likes
Related Questions
There are two copper wires of length ratio 1:2 that have their cross-sectional areas in the ratio 1:4. What will be the ratio of their:
(a) resistances?
(b) specific resistances?AB is an object and PQ is its real, inverted image. The lengths of AB and PQ are equal. This is possible when a lens is present between the object and its image.
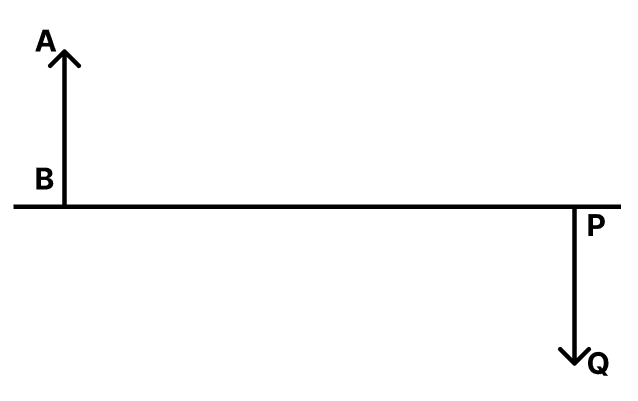
(a) What kind of lens is used here? Also, state the position of the lens and where it is to be placed.
(b) Name a device where this lens action is used.
Soumya took two right circular cones of the same vertical height. One of the two cones is a solid one, while the other is hollow from inside. By measuring the cross-sectional areas of the cones and from the knowledge of symmetry, by using suitable formulas, he found the positions of the centre of gravity of both cones. He found that the difference is about 1.5 cm. What is the vertical height of the two cones?
Seismic waves have different frequencies. During earthquakes, why are short-length buildings more prone to damage caused by high-frequency seismic waves?