Chemistry
Water acts as a universal solvent because it has a unique property to dissolve various organic and inorganic compounds. Its dielectric constant is high, which is about 80 at 20°C. All this is due to its polar nature.
(a) What properties of water make it an exceptional solvent compared to other solvents like ethanol, carbon tetrachloride, benzene, etc.
(b) Explain the environmental consequences of water's "universal solvent" property, particularly regarding pollution.
Answer
(a) Water has following properties that make it an exceptional solvent :
- Water is an exceptional solvent because its molecules are polar in nature. The oxygen atom has a partial negative charge and the hydrogen atoms have partial positive charges. Due to this polarity, water molecules can attract different types of particles and help them dissolve.
- Water also has a high dielectric constant. Therefore, it reduces the electrostatic force of attraction between positive and negative ions and dissolves many inorganic compounds. Hence, water can dissolve a wide range of solids, gases and liquids, forming aqueous solutions.
(b) Water’s property of being a universal solvent also makes it easily polluted. It can dissolve or carry harmful substances such as sewage, industrial wastes, pesticides, fertilisers and toxic chemicals. As a result, water may become unsafe for drinking and can cause diseases such as diarrhoea, typhoid, cholera and dysentery. Long-term consumption of chemically polluted water may also cause toxicity, cancer or organ damage.
Related Questions
Srishti needs to dry ammonia gas in the laboratory. She knows the following drying agents:
- Solid calcium chloride
- Concentrated sulphuric acid
- Solid calcium oxide
- Phosphorus pentoxide
However, she chooses to use solid calcium oxide.
Answer the following questions:
(a) Why is solid calcium oxide preferred over other drying agents for drying ammonia gas?
(b) Why is concentrated sulphuric acid not used to dry ammonia gas?
(c) Concentrated sulphuric acid is a good dehydrating agent. Explain.
(d) State one difference between a drying agent and a dehydrating agent.
A solution of a solid in a liquid (such as water) can be prepared by adding the solid to water with constant stirring at a particular temperature. If the addition process is continued, a stage is ultimately reached in the dissolution process when no more of the solid dissolves. The solution at this stage is said to be saturated. The solubility of a solute is always expressed with respect to the saturated solution. It may be defined as the maximum amount of the solute that can be dissolved in 100 g of the solvent to form a saturated solution at a given temperature.
The solution becomes unsaturated if the temperature is increased. In case the temperature is decreased, the solution becomes supersaturated and crust of the solute gets deposited on the surface.(a) A student studied the solubility of three substances in water at different temperatures and collected the following data.
Temperature (°C) 286 300 320 350 Substance dissolved Solubility X 20 24 26 30 Y 30 36 48 54 Z 24 20 18 14 The correct order of the solubilities of these substances in water is :
(i) Y > X > Z
(ii) X > Y > Z
(iii) Z > X > Y
(iv) Y > Z > X(b) 10 g of a solute is dissolved in 250 g of a solvent. The solubility of the solute is:
(i) 4 g
(ii) 40 g
(iii) 10 g
(iv) 20 g(c) A saturated solution becomes unsaturated by :
(i) Heating the solution
(ii) Cooling the solution
(iii) Adding more of the solute
(iv) Adding more of the solvent(a) (i) and (iii)
(b) (ii) and (iii)
(c) (i) and (iv)
(d) (iii) and (iv)(d) Solubility of a solute in a solvent may be defined as:
(i) Dissolution of 1 g of solute in one kg of solvent at a given temperature.
(ii) Dissolution of 1 g of solute in 100 g of the solvent at a given temperature.
(iii) Dissolution of 1 g of solute in 100 mL of the solvent at a given temperature.
(iv) The maximum amount of solute which can be dissolved in 100 g of the solvent to form a saturated solution at a given temperature.A group of students took an old shoe box and covered it with a black paper from all sides. They fixed a source of light (a torch) at one end of the box by making a hole in it and made another hole on the other side to view the light. They placed a milk sample contained in a beaker/tumbler in the box as shown in the figure. They were amazed to see that milk taken in the tumbler was illuminated. They tried the same activity by taking a salt solution but found that light simply passed through it.
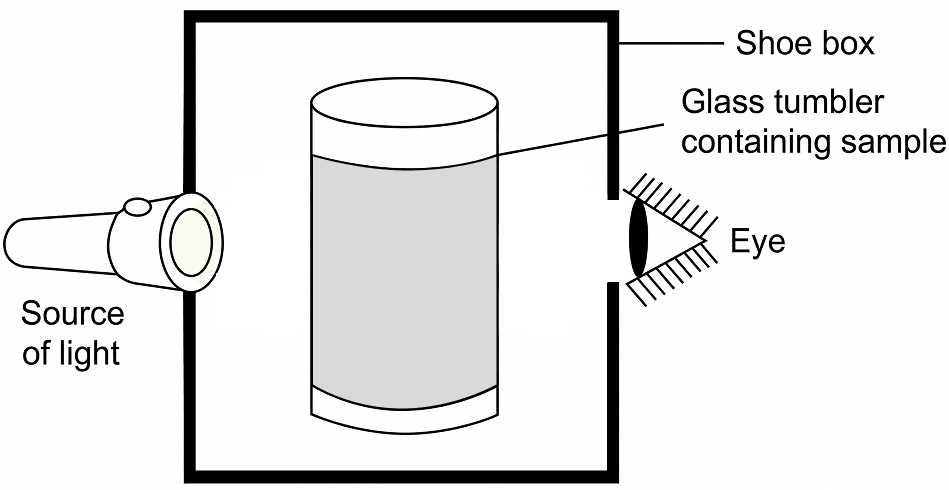
(a) Explain why the milk sample was illuminated. Name the phenomenon involved.
(b) Same results were not observed with a salt why ?
(c) Can you suggest two more solutions which would show the same effect as shown by the milk solution ?
Name some physical and chemical processes used to make river water potable.