Chemistry
Related Questions
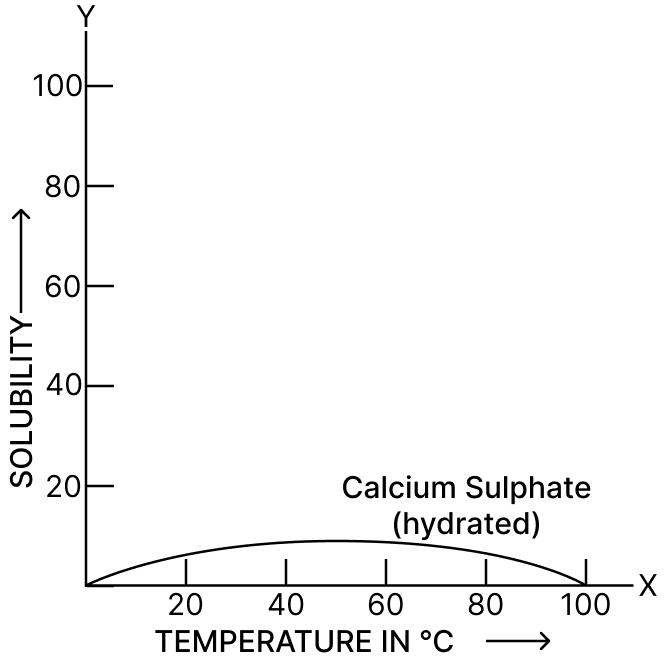
Give three factors which affect the solubility of a solid solute in a solvent.
(a) If you are given some copper sulphate crystals, how would you proceed to prepare its saturated solution at room temperature?
(b) How can you show that your solution is really saturated?
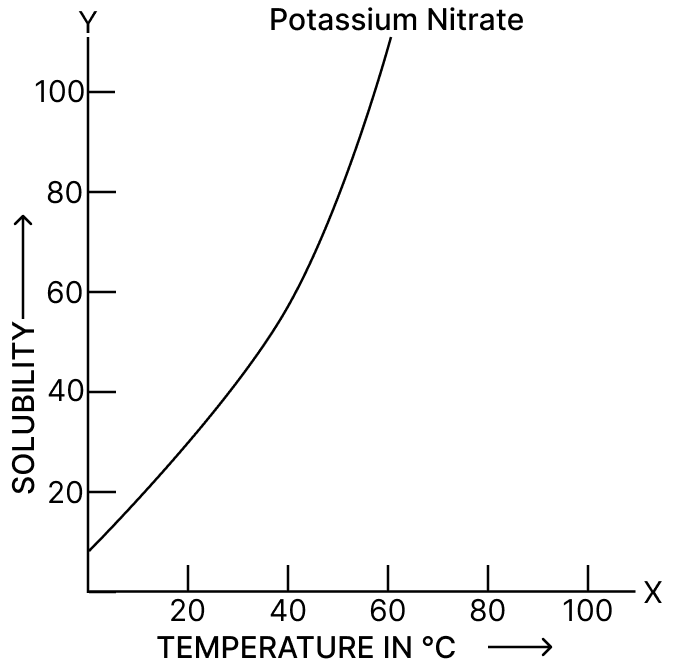
Solubility of NaCl at 40°C is 36.5 g. What is meant by this statement
Which test will you carry out to find out if a given solution is saturated or unsaturated or supersaturated?