With respect to the Hall Heroult process related to the extraction of aluminium, justify the following:
(a) Powdered Coke is sprinkled over the electrolytic mixture undergoing electrolytic reduction.
(b) Graphite anodes are continuously replaced during the electrolysis.
(c) Cryolite and fluorspar must be added to the electrolytic mixture.
Answer
(a) The layer of powdered coke is sprinkled over the electrolytic mixture as:
- It prevents burning of carbon electrodes in air at the emergence point from the bath.
- It minimizes or prevents heat loss by radiation.
(b) The graphite anodes are continuously replaced during the electrolysis because:
- The oxygen evolved at the anode escapes as a gas or reacts with the carbon anode.
- The carbon anode is thus oxidized to carbon monoxide which either burns giving carbon dioxide or escapes out through an outlet.
2C + O2 ⟶ 2CO
2CO + O2 ⟶ 2CO2
(c) Cryolite and fluorspar are added to the electrolytic mixture because their addition:
- lowers the fusion point of the mixture i.e., mixture fuses around 950°C instead of 2050°C.
- enhances the mobility of the fused mixture by acting as a solvent for the electrolytic mixture.
- enhances the conductivity of the mixture since, pure alumina is almost a non-conductor of electricity.
Write complete and balanced equations for the reactions occurring in the following cases:
(a) Passing dry ammonia gas over heated lead oxide placed in a combustion tube to produce a silvery grey metal.
(b) When concentrated nitric acid is reacted with zinc to produce a reddish-brown gas.
(c) When concentrated sulphuric acid oxidises sulphur to produce a gas which turns acidified potassium dichromate paper green.
Answer
(a) NH3 + 3PbO ⟶ 3Pb + 2H2O + 2NO2
(b) Zn + 4HNO3 ⟶ Zn(NO3)2 + 2H2O + 2NO2
(c) 2H2SO4 + S ⟶ 3SO2 + 2H2O
Give balanced equations for the conversions A, B and C.
Answer
(A) Zn + 2HCl ⟶ ZnCl2 + 2HCl
(B) ZnCl2 + 2NaOH ⟶ Zn(OH)2 + 2NaOH
(C) Zn(OH)2 + H2SO4 ⟶ ZnSO4 + 2H2O
Rohan wants to electroplate a spoon with nickel.
(a) To which electrode should he connect the article to be electroplated?
(b) Write the equation for the reaction that will occur at the cathode.
(c) What should the anode be made up of?
Answer
(a) The article to be electroplated is placed is made as cathode. This is because metal cations from the electrolyte will move toward the cathode and get reduced and deposited on the spoon.
(b) Ni2+ + 2e- ⟶ Ni
(c) The anode should be made of pure nickel metal. During electroplating, nickel atoms from the anode dissolve into the solution as Ni2+ ions, maintaining a steady concentration of nickel ions in the electrolyte.
Amit found that 30g of a gas occupied 1000 c.c at STP.
(a) What will the gram molecular weight and the vapour density of this gas be?
(b) How many molecules of this gas will be present in 44.8 l of it?
Answer
Given,
Mass of gas = 30g
Volume = 1000 c.c = 1dm3 = 1 liter
We know one mole of gas at STP occupies 22.4 l
1 litres of gas at S.T.P. has mass = 30 g
∴ 22.4 litre (molar volume) has mass
= x 22.4
= 672 g/mol
Vapour density =
=
= 336
(b) 22.4 litres of the gas contains 6.023 x 1023 molecules
So, 44.8 litres of the gas contain 2 x 6.023 x 1023
= 12.046 x 10 23 molecules
An element X combines with oxygen to form an oxide X2O3. This oxide is a good conductor of electricity and can be reduced to its metal only by electrolysis.
(a) Write the equation for the reaction formed when the oxide (X2O3) combines with hydrochloric acid.
(b) How many valence electrons are present in the outermost shell of X?
(c) Will element X undergo oxidation or reduction?
Answer
Element X is a metal, and its oxide is X2O3 so X must have a valency of 3 (X3+). The oxide is a good conductor of electricity. Since it requires electrolysis to extract X, it must be a very reactive metal.
(a) The oxide (X2O3) is reacting with hydrochloric acid, so we can say the oxide is ionic and basic in nature. The reaction will give salt and water.
X2O3 + 6HCl ⟶ 2XCl3 + 3H2O
(b) Since X forms X3+, it must lose 3 electrons, meaning it has 3 valence electrons.
(c) To form X3+, element X must lose 3 electrons and loss of electron is oxidation.
Observe the reactions given below and answer the following questions:
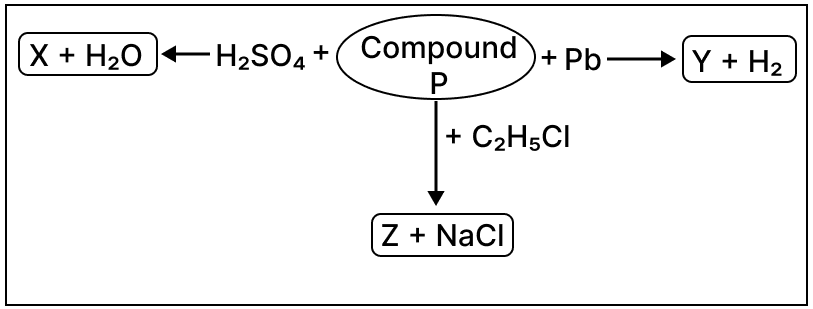
(a) Identify compound P.
(b) Give the chemical formula of Z.
(c) Write the reaction taking place between the identified compound P and sulphuric acid.
(d) Name compound Y.
Answer
(a) NaOH
(b) C2H5OH
(c) 2NaOH + H2SO4 ⟶ Na2SO4 + 2H2O
(d) Sodium plumbite
Study the information given in the table below and answer the questions that follow. (Note- the letters do not represent the actual symbols of the elements)
| Element | Electronic configuration | Ionisation energy kjmol-1 |
|---|---|---|
| X | 2,2 | 900 |
| Y | 2,8,2 | 738 |
| Z | 2,8,8,2 | 590 |
(a) Explain why element X has highest ionisation energy.
(b) To which period does Z belong?
(c) Draw the electron dot structure of the compound formed between Z and oxygen.
Answer
(a) Element X has the highest ionisation energy because it has the smallest atomic size, fewer electron shells, and its valence electrons experience a stronger attraction to the nucleus.
(b) Z has the configuration of 2,8,8,2, electrons occupying K,L,M and N shells. Hence, Z belongs to Period 4.
(c) The electron dot structure of the compound formed between Z and oxygen is shown below:
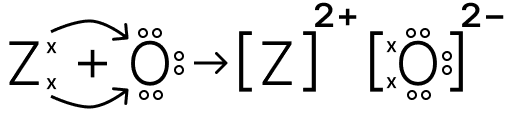
A student prepared a Potassium sulphite solution in the lab and added few drops of barium nitrate solution to it. He observed a white precipitate being formed in the test tube. On addition of dilute hydrochloric acid to the white precipitate and mixing it, he observed that the precipitate disappeared.
(a) Name the white precipitate.
(b) Write a balanced chemical equation for the reaction between dilute hydrochloric acid and the white precipitate.
(c) Name the gas evolved in the above reaction.
Answer
(a) When the student mixed potassium sulphite solution (K2SO3) with barium nitrate (Ba(NO3)2), the white precipitate of Barium sulphite (BaSO3) is formed.
K2SO3 + Ba(NO3)2 ⟶ BaSO3 + 2KNO3
(b) When white precipitate of BaSO3 reacts with dilute HCl, it dissolves, forming barium chloride (BaCl2), water, and sulphur dioxide (SO2) gas.
BaSO3 + 2HCl ⟶ BaCl2 + H2O + SO2 ↑
(c) From the above reaction, the gas produced is sulphur dioxide (SO2).
The empirical formula of a hydrocarbon is C2H3. The hydrocarbon has a relative molecular mass of 54. (At wt: H = 1, C = 12)
(a) What is the molecular formula of the hydrocarbon?
(b) Draw the structural formula of the hydrocarbon.
(c) Give the general formula of the hydrocarbon.
Answer
(a) Empirical formula = C2H3
Relative molecular mass = 54
∴ Molecular formula = n[E.F.] = 2[C2H3] = C4H6
(b) The structural formula of butyne is shown below:
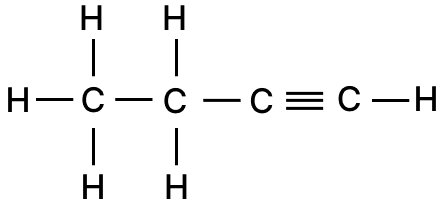
(c) The hydrocarbon C4H6 is an alkyne, and the general formula for alkyne is CnH2n-2.
Study the given figure and answer the given questions:
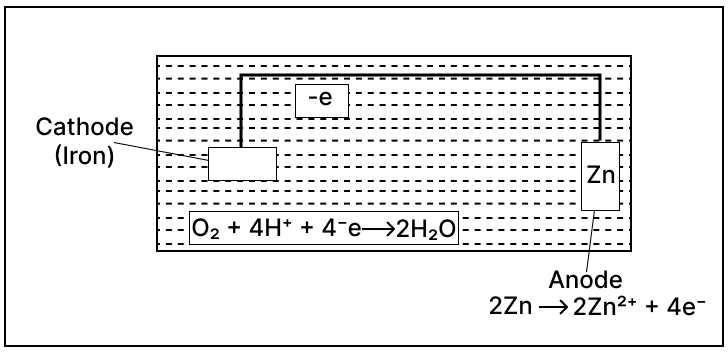
(a) Identify the application of electrolysis demonstrated above.
(b) Which metal is protected in the above process?
(c) Why should the metal be protected?
Answer
(a) Electroplating
(b) Iron
(c) Iron tools and instruments are often electroplated with zinc to protect against rusting.
Nita's father bought a basket of ripe mangoes. While opening it she found a small sachet containing a white crystalline powder along with the mangoes. She was told that it is a chemical that releases a gas when it comes in contact with moisture, that induces ripening of fruits.
(a) Name the chemical powder in the sachet.
(b) Name the gas.
(c) Give a balanced chemical equation for the reaction that results in the evolution of this gas.
Answer
(a) Calcium carbide (CaC2)
(b) Acetylene/Ethyne.
(c)
Atomic number of element M is 12 and it forms an ionic compound with element L.
(a) Which of the following atomic numbers will match L?
i. 14
ii. 10
iii. 8
(b) What is the name given to the members of the group to which element M belongs?
(c) Draw the electron dot structure of the compound formed between M and L.
Answer
Atomic number of element M = 12
Electronic configuration of element M = 2, 8, 2
∴ Element M is a metal with valency 2, it will form M2+ ion
(a)
| Atomic Number | Configuration | Evaluation |
|---|---|---|
| 14 | 2, 8, 4 | Tends to share electrons and forms covalent bonds |
| 10 | 2, 8 | Noble gas – inert, does not form ions |
| 8 | 2, 6 | Non-metal, gains 2 electrons to form ionic compounds |
The correct match for L will be (iii), because M can form an ionic compound with L (M2+ + L2- = ML)
(b) As element M contains 2 valenced electrons, it belongs to Group 2 of the Periodic Table and group 2 elements are called Alkaline Earth Metal.
(c) The electron dot structure of the compound formed between M and L is shown below:
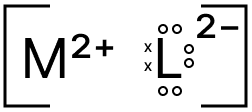
When two dry gases, oxygen and X, are passed over heated platinum, reddish-brown fumes are seen in the receiving flask, as shown in the figure.
(a) Name the gas X.
(b) Give equation(s) for the reaction(s) that resulted in the formation of brown fumes.
Answer
(a) The gas X is Ammonia (NH3).
(b) 4NH3 + 5O2 4NO↑ + 6H2O + Δ
2NO + O2 ⟶ 2NO2 [brown gas]