Some general rules for the solubility of salts in water are listed.
- Carbonates are insoluble (except ammonium carbonate, potassium carbonate and sodium carbonate).
- Chlorides are soluble (except lead (II) chloride and silver chloride).
- Nitrates are soluble.
- Sulphates are soluble (except barium sulphate, calcium sulphate and lead (II) sulphate).
Which substances produce an insoluble salt when aqueous solutions of them are mixed?
Justify your answer.
(a) Copper nitrate and magnesium chloride
(b) Zinc chloride and ammonium nitrate
(c) Silver nitrate and zinc chloride
(d) Potassium carbonate and sodium sulphate
Answer
The salts formed and their solubility when the given reactants are mixed is listed in the table below:
| Reactant 1 | Reactant 2 | Products | Solubility |
|---|---|---|---|
| Copper nitrate | Magnesium chloride | CuCl2 Mg(NO3)2 | Soluble Soluble |
| Zinc chloride | Ammonium nitrate | Zn(NO3)2 NH4Cl | Soluble Soluble |
| Silver nitrate | Zinc chloride | AgCl Zn(NO3)2 | Insoluble Soluble |
| Potassium carbonate | Sodium sulphate | K2SO4 Na2CO3 | Soluble Soluble |
From the table, we can see that silver nitrate and zinc chloride form silver chloride (AgCl) which is an insoluble precipitate, while for all others, no precipitate is formed.
Ammonia gas is passed into water as shown below:
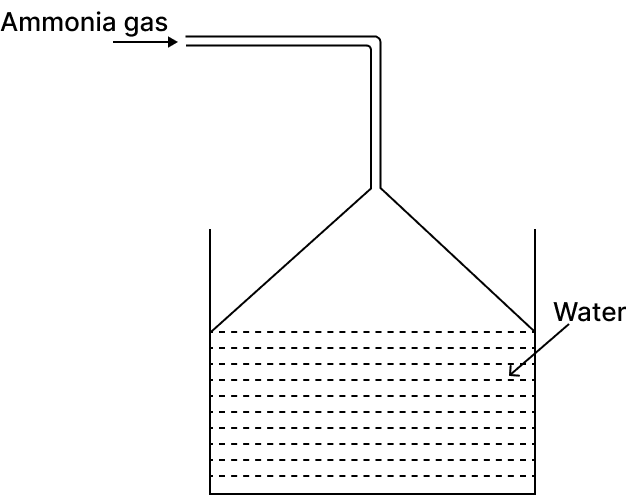
(a) When a red litmus paper was dropped into the resulting solution, it turned blue. Which ions in the solution would have resulted for the colour change in the litmus paper?
(b) Why is the funnel kept in an inverted position?
Answer
(a) The red litmus paper turned blue, which indicates that the solution is basic (alkaline) in nature. Hydroxyl ions (OH-) are responsible for turning red litmus paper blue.
(b) To prevent back suction of water and to increase area of absorption of Ammonia gas.
In the Haber’s process, the optimum yield of ammonia is obtained when a temperature of 450°C - 500°C, a pressure of 200 atmospheres, an iron catalyst and promoter molybdenum are used.
N2 + 3H2 2NH3 + heat
How and why would the yield of ammonia be affected if the temperature was raised to 600°C?
Answer
Raising the temperature to 600°C would decrease the yield of ammonia because the forward reaction is exothermic, and increasing temperature shifts the equilibrium toward the endothermic reverse reaction, reducing ammonia formation.
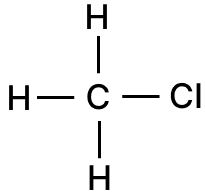
Give the structural formula and the name of the organic product formed when equal volumes of methane and chlorine react together.
Answer
Chloromethane or methyl chloride
Complete combustion of one mole of a hydrocarbon produced four moles of carbon dioxide and four moles of water only.
(a) Write the equation for the combustion reaction.
(b) Draw the structure of the hydrocarbon.
Answer
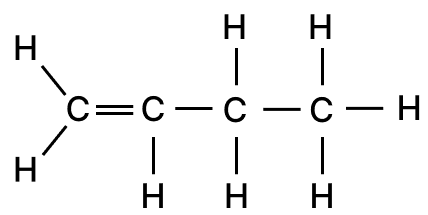
(a) Complete combustion of butene (C4H8) produces four moles of carbon dioxide and four moles of water only.
C4H8 + 6O2 ⟶ 4CO2 + 4H2O
(b) Structural formula of butene (C4H8) is shown below:
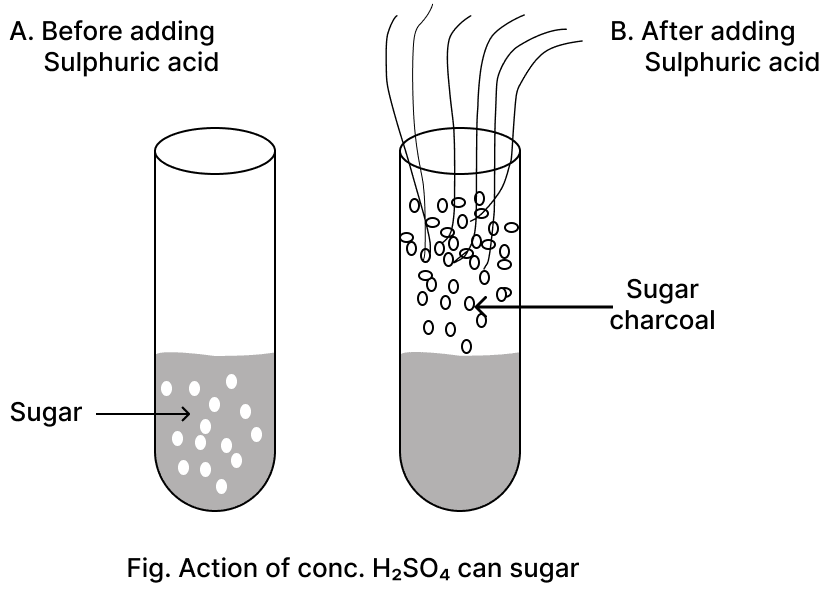
To the acid prepared by the contact process, Barium chloride solution is added. State one observation and write an equation for the reaction that occurs.
Answer
When barium chloride solution is added to the acid prepared by the contact process i.e., sulphuric acid (H2SO4), a white precipitate of barium sulphate (BaSO4) is formed, which is insoluble in water.
H2SO4 + BaCl2 ⟶ BaSO4 + 2HCl
Platinum catalyst is used in the catalytic oxidation of ammonia.
(a) Write an equation for the reaction that occurs in the above case.
(b) Why does the platinum continue to glow even after the heating is discontinued?
Answer
(a) Equation for the catalytic oxidation of ammonia is given below:
(b) The platinum (catalyst) continues to glow even after the heating is discontinued because the catalytic oxidation of ammonia is an exothermic reaction.
With reference to the reaction occurring in the given figure: -
(a) Write an equation for the reaction.
(b) State the property of sulphuric acid exhibited in the above case.
Answer
(a)
(b) Conc. H2SO4 acts as dehydrating agent. It readily removes chemically combined water molecules.
Brown ring test is used for the identification of nitrate ions.
(a) Why is freshly prepared Ferrous sulphate solution used in the above test?
(b) What is the chemical name of the brown ring?
Answer
(a) A freshly prepared ferrous sulphate solution is used because on exposure to the atmosphere, it is oxidised to ferric sulphate, which will not give the brown ring test.
(b) FeSO4.NO
[Nitroso Ferrous sulphate, a brown compound]
Ravi heated 367.5 g of KClO3 in a test tube. The decomposition of potassium chlorate took place according to the equation.
2KClO3 ⟶ 2KCl + 3O2
Find:
(a) the volume of the colourless and odourless gas liberated during the experiment.
(b) the weight of the residue left behind in the test tube.
Answer
Given,
Mass of KClO3 = 367.5 g
Molar mass of KClO3 = 39 (K) + 35.5 (Cl) + 3×16 (O) = 122.5 g/mol
Molar mass of KCl = 39 (K) + 35.5 (Cl) = 74.5
(a) 245 g of KClO3 produces 67.2 lit. of O2
∴ 367.5g of KClO3 will give,
= 67.2 x = 100.8 l of O2
∴ The volume of the colourless and odourless gas O2 liberated during the experiment is 100.8 liters.
(b) 245 g of KClO3 produces 149 g of KCl
∴ 367.5g of KClO3 produces,
= 149 x = 223.5g of KCl
∴ The weight of the KCl left behind as a residue in the test tube is 223.5g.
For construction work the alloy of Aluminium i.e., Duralumin is used rather than pure Aluminium. Give two valid reasons.
Answer
Two reasons for using Duralumin instead of Aluminium is:
- Duralumin is light and strong, while aluminium is light and weak. It is also unaffected by moist air. Duralumin has a strength up to six times greater than pure aluminium.
- It is corrosion-resistant and has high tensile strength.
Ram took 5 moles of carbon atoms in a container, and Krish took 5 moles of sodium atoms in another container of the same volume.
(a) Whose container is heavier?
(b) Which container has a larger number of atoms?
Answer
(a) Carbon (C): Atomic mass = 12 g/mol
Mass = 5 moles x 12 g/mol = 60 g
Sodium (Na): Atomic mass = 23 g/mol
Mass = 5 moles x 23 g/mol=115 g
∴ Krish’s container is heavier
(b) 1 mole = 6.022 x 1023 atoms
Both the containers have 5 moles of substance.
5 moles x 6.022 x 1023 = 3.011 x 1024 atoms
∴ Both the containers have the same number of atoms.
A, B and C are three elements which undergo chemical reactions according to the following equations:
A2O3 + 2B ⟶ B2O3 + 2A
3CSO4 + 2B ⟶ B2(SO4)3 + 3C
3CO + 2A ⟶ A2O + 3C
Answer the following questions:
(a) Which element is the most reactive?
(b) Which element is the least reactive?
Answer
(a) Most reactive element is B
(b) Least reactive element is C
Explanation:
Above reactions are displacement reaction. In first and second reactions B is displacing A and C from their oxide and sulphate respectively. In third reaction, A displaces C from its oxide. Considering all three reaction, B is more reactive than A and C.
C is less reactive, and is not displacing either A and B in above reactions.
PQ2 is a hard crystalline solid having high melting and boiling points. It is a good conductor of electricity in both molten and aqueous forms.
(a) The conductivity of PQ2 is due to the presence of free ............... (ions, molecules, electrons)
(b) During electrolysis of aqueous PQ2, if thickening of the cathode and thinning of the anode is observed, the anode material will be ............... . (graphite, metal P)
Answer
(a) The conductivity of PQ2 is due to the presence of free ions.
(b) During electrolysis of aqueous PQ2, if thickening of the cathode and thinning of the anode is observed, the anode material will be metal P.
Explanation:
PQ2 is an ionic compound made of metal P and nonmetal Q, where P is likely a divalent metal cation (P2+)
(a) Ions, ionic compounds conduct electricity in molten or aqueous forms due to the movement of ions.
(b) Thickening of the cathode means metal P2+ ions gain electrons (reduction) and deposit as solid metal P. Thinning of the anode means anode is dissolving, which only happens if the anode is made of the same metal (P).
If the anode were graphite, it would not dissolve, but might produce gas instead. So, the anode material must be metal P
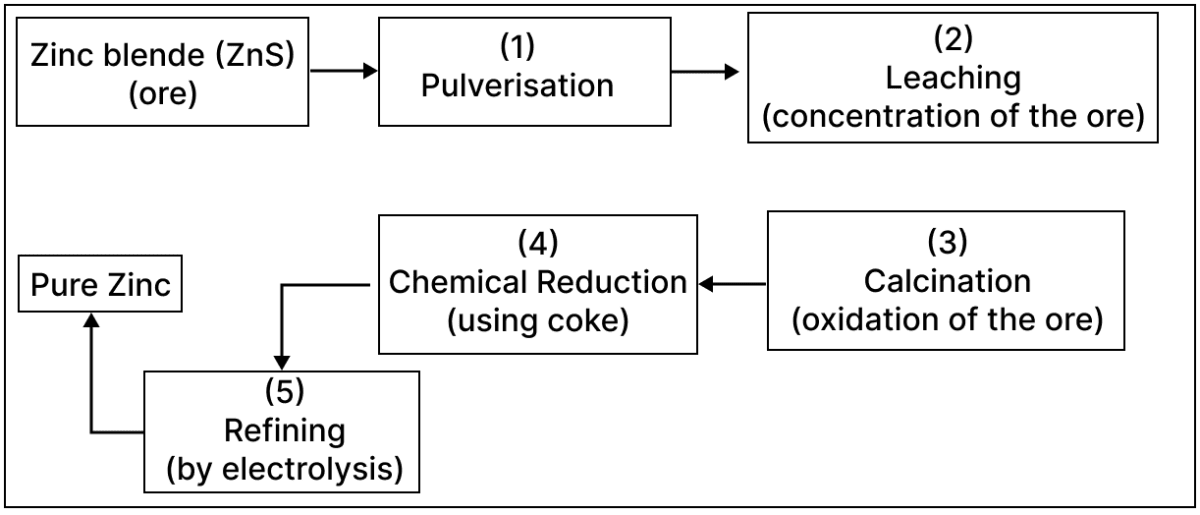
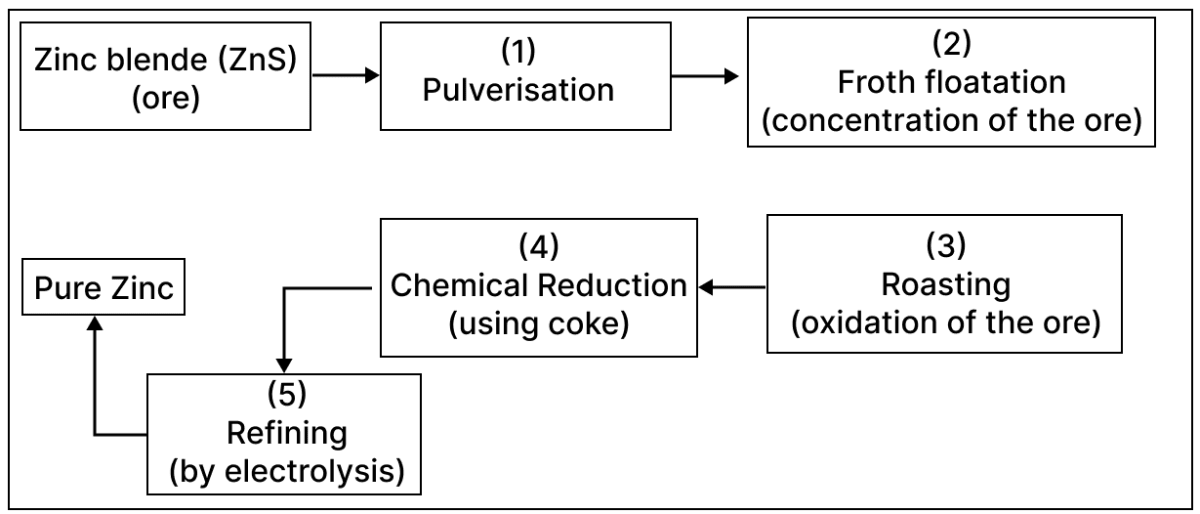
A student was asked to draw the flowchart for the extraction of zinc from zinc blende based on the principles of Metallurgy. What he drew is given below.
2 steps out of the 5 were incorrect. Identify and correct them.
Answer
The incorrect steps are:
- Step 2: Froth floatation instead of leaching.
- Step 3: Roasting instead of calcination.
The corrected flowchart is shown below:
Given below is the representation of the conversion of ethene to a saturated hydrocarbon X, where ‘a’ stands for the catalyst.
C2H4 X
(a) Identify ‘a’.
(b) Give the complete chemical equation for the conversion of C2H4 to X.
Answer
(a) Nickle
(b) The complete chemical equation for the conversion of C2H4 to C2H6 (Ethane) is shown below:
The description of an organic compound is as follows:
(a) Molecular formula is C3H8O.
(b) Functional group is attached to the first carbon atom.
(c) Reacts with sodium at room temperature with brisk effervescence, releasing hydrogen gas.
Identify the compound and draw its structure.
Answer
The compound is Propan-1-ol.
The structure of the compound is given below:
