Physics
(a)
- Which element is used in the lining of the special aprons worn by workers in nuclear power plants?
- Why is this element preferred?
(b) emits a nuclear radiation which does not alter the mass number but is deflected by a magnetic field.
- Name the type of nuclear radiation emitted by .
- Write the equation for this radioactive decay.
Radioactivity
3 Likes
Answer
(a)
- Lead is used in the lining of the special aprons worn by workers in nuclear power plants.
- Lead is preferred because it is a dense metal and absorbs harmful nuclear radiations effectively.
(b)
- The radiation that does not change the mass number but is deflected by a magnetic field is beta (β) radiation.
Hence, beta (β) radiation is emitted by . - Radioactive decay equation:
In this decay, a beta particle (electron) is emitted, the atomic number increases by 1, while the mass number remains unchanged.
Answered By
1 Like
Related Questions
The reverse side of a three-pin plug with incorrect connection of wires is shown in the diagram below.

(a) Identify the fault in the above connection.
(b) Mention a risk factor involved, if the user operates the appliance without correcting it.
(c) Will the appliance function in the present situation? (Yes or No)
In the combinations of resistors shown below. calculate:
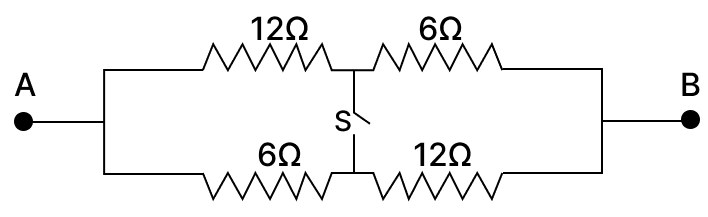
(a) the resistance across AB when the switch S is open.
(b) the resistance across AB when the switch S is closed.
An electric iron rated 1100 W, 220 V is operated for 5 hours.
Calculate:
(a) the minimum rating of the fuse required.
(b) the energy consumed in kWh.
(c) the cost of the energy consumed, if the rate is ₹ 10 per unit.
When the magnet as shown in the diagram, is moved towards the coil at a speed of 5 m s-1, the galvanometer shows a certain deflection to the right.
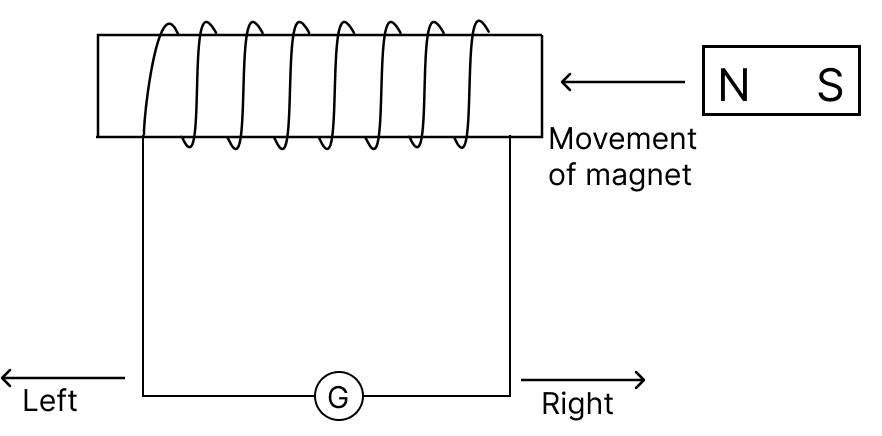
How will the direction and magnitude of deflection change when the coil also moves with a speed of 5 m s-1:
(a) in the direction of the motion of the magnet?
(b) in the opposite direction of the motion of the magnet?