Chemistry
A student takes Cu, Al, Fe and Zn strips, separately in four test tubes labeled as I, II, III and IV respectively. He adds 10 ml of freshly prepared ferrous sulphate solution to each test tube and observes the colour of the metal residue in each case.
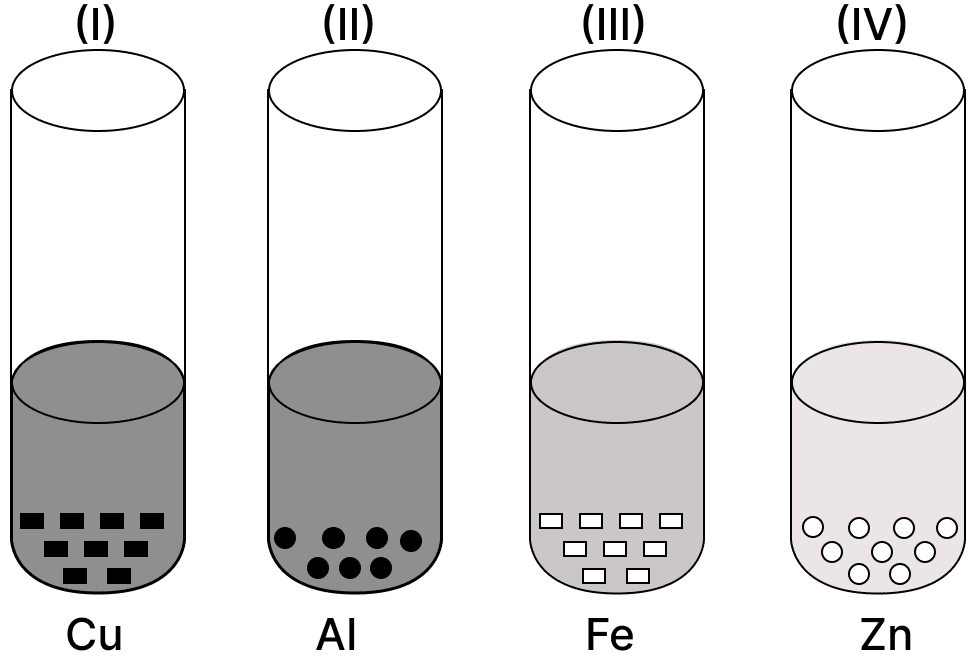
He would observe a black residue in the test tubes:
- (I) and (II)
- (I) and (III)
- (II) and (III)
- (II) and (IV)
Related Questions
Aqueous lead (II) nitrate can be distinguished from aqueous zinc nitrate by adding any of the following solution in excess, except:
- aqueous potassium chloride
- aqueous sodium sulphate
- dilute sulphuric acid
- sodium hydroxide solution
Which of the following about oxides is correct?
- A basic oxide is an oxide of a non-metal
- Acidic oxides contain ionic bonds
- Amphoteric oxides contain a metal
- Basic oxides are always gases
Electroplating steel objects with silver involves a three-step process.
Step 1 — A coating of copper is applied to the object.
Step 2 — A coating of nickel is applied to the object.
Step 3 — The coating of silver is applied to the object.
(a) A diagram of the apparatus used for step 1 is shown.

The chemical process taking place on the surface of the object is
Cu2+(aq) + 2e- ⟶ Cu (s)
What is the observation seen on the surface of the object?Explain why the concentration of copper ions in the electrolyte remains constant throughout step 1.
(b) Give two changes which would be needed in order to coat nickel onto the object in step 2.
(c) Write down the reaction taking place at the positive electrode during step 3.
Match the following Column A with Column B.
Column A Column B Aluminium Covalent compound Sulphuric acid Carbonate ore Calcination Hall Heroult’s process Calcium Chloride Contact Process Carbon tetrachloride Electrovalent compound