Chemistry
An ore on being heated in air forms sulphurous anhydride. Write the process used for the concentration of this ore.
Related Questions
Give equations for the reduction of :
(a) Iron (II) oxide
(b) Iron (III) oxide
(c) Lead (II) oxide
(d) Zinc oxide
(a) Some metallic oxides can be reduced by hydrogen, carbon, carbon monoxide and some cannot. Explain.
(b) Write balanced equation for the reduction of copper (II) oxide by hydrogen.
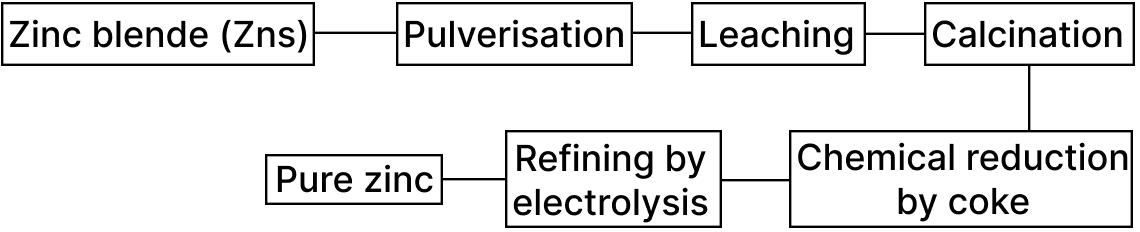
The following flow chart shows the process of extraction of zinc. Out of the 5 steps, 2 steps are incorrect. Identify and correct them.
State why aluminium is extracted from it's oxide by electrolysis while copper, lead, iron by reducing agents and mercury and silver by thermal decomposition.