Chemistry
Assertion (A): In a redox reaction, the electron gaining species acts as a reducing agent.
Reason (R): Oxidation and reduction reactions occur simultaneously in redox reaction.
- Both A and R are true and R is the correct explanation of A.
- Both A and R are true but R is not the correct explanation of A.
- A is true but R is false.
- A is false but R is true.
Chemical Reaction
13 Likes
Answer
A is false but R is true.
Explanation — The electron gaining species is being reduced, so it acts as an oxidizing agent, not as a reducing agent. The reducing agent is the one that loses electrons. Hence the assertion (A) is false.
A redox reaction is a chemical reaction where both oxidation and reduction occur simultaneously. Hence the reason (R) is true.
Answered By
8 Likes
Related Questions
See the diagram given below and answer the following questions:
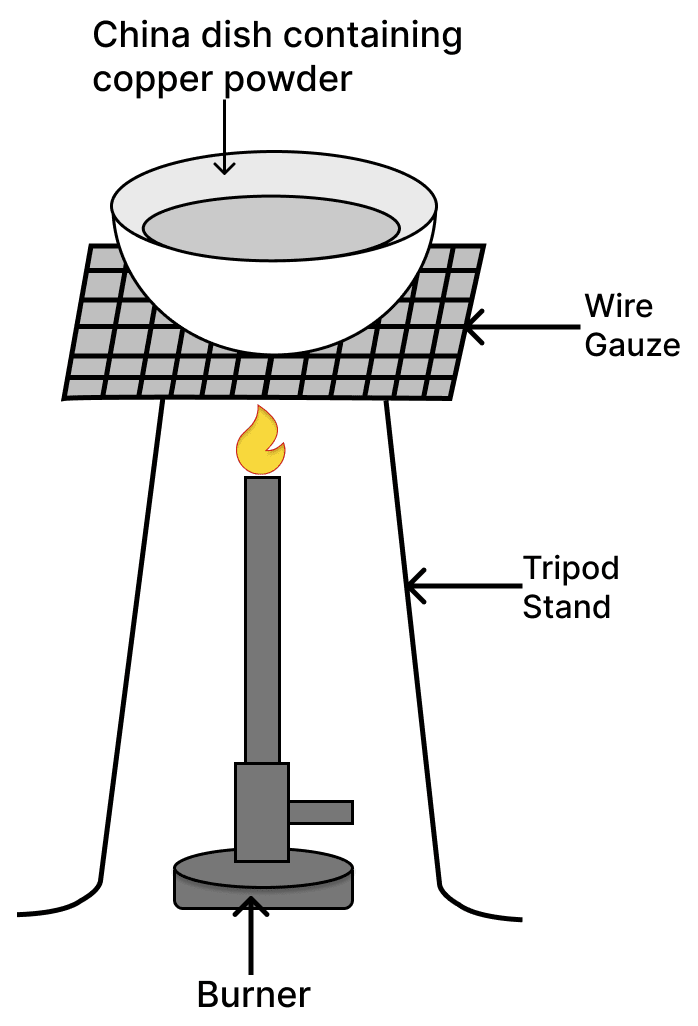
(a) Write the chemical reaction involved in the process.
(b) Mention the colour of :
(i) copper powder and
(ii) the substance formed after heating it.(c) Can we reverse the above reaction ? If yes, write the equation for the reverse reaction and state the substance that undergoes oxidation and the substance that undergoes reduction.
Assertion (A): Zinc can displace copper from aqueous copper sulphate solution.
Reason (R): Copper is placed above zinc in the reactivity series.
- Both A and R are true and R is the correct explanation of A.
- Both A and R are true but R is not the correct explanation of A.
- A is true but R is false.
- A is false but R is true.
Assertion (A): The decomposition of vegetable matter into compost is an endothermic reaction.
Reason (R): Decomposition reactions involve the breakdown of a single reactant into simpler products.
- Both A and R are true and R is the correct explanation of A.
- Both A and R are true but R is not the correct explanation of A.
- A is true but R is false.
- A is false but R is true.
Assertion (A): The decomposition of silver bromide is utilized in black and white photography.
Reason (R): Light provides energy for this endothermic reaction.
- Both A and R are true and R is the correct explanation of A.
- Both A and R are true but R is not the correct explanation of A.
- A is true but R is false.
- A is false but R is true.