Physics
(a) Convert to SI unit: 1 J/g °F
(b) Why does 1 g of water at 0°C have 336 J more heat energy than 1 g of ice at 0°C?
Calorimetry
16 Likes
Answer
(a) 1 J/g °F = 1800 J / (kg K)
(b) 336J of energy is used to increase the potential energy due to an increase in intermolecular separation, so the latent heat of ice is 336 J.
Explanation:
(a) ∵ 1 gram = 0.001 kg
Now convert:
(b) At 0 °C, ice and water are at the same temperature, but not at the same energy state. To convert 1 g of ice at 0 °C into 1 g of water at 0 °C, it must absorb latent heat.
This latent heat of fusion for water is: Lf = 336 J/g
So, to melt 1 g of ice: Q = mL = 1 g x 336 J/g = 336 J
Thus, 1 g of water at 0 °C has 336 J more energy than ice at the same temperature because that’s the energy absorbed during the phase change.
Answered By
10 Likes
Related Questions
Study the diagram and answer the questions that follow.
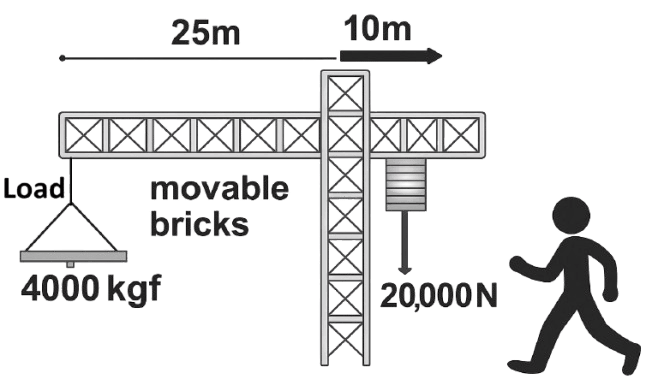
(a) In the diagram, is the worker attempting to raise or lower the load?
(b) Justify your answer to (a) with the necessary calculation.
The graph shows load against effort for a lever with load and effort on the same side of the fulcrum.
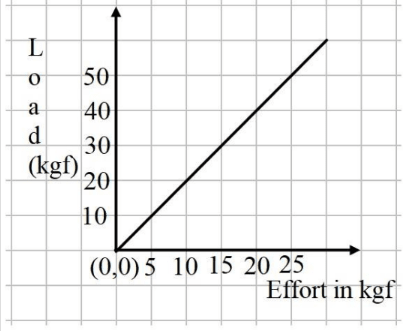
(a) Which feature of the load and effort graph must be calculated to determine mechanical advantage?
(b) Which class does this lever belong to?
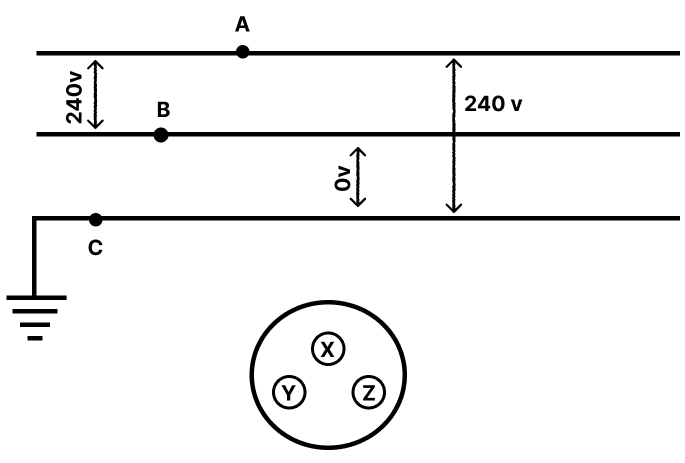
Match the columns by choosing the body part and corresponding mechanical lever of the same class.
Human body part Mechanical lever i. Nodding head (a) Bottle Opener ii. Lifting body weight on your toes. (b) Tongs (c) See-Saw Redraw the diagram by linking points A, B, and C to points X, Y, and Z on the socket.