Chemistry
The electronic configuration of X is 2,8,6. It gains 'Y’ electrons into its valence shell to attain the nearest noble gas electronic configuration and gets converted to an ion Z. X, Y, and Z, respectively, are:
- Sodium, one, electropositive
- Beryllium, two, electronegative
- Oxygen, six, electronegative
- Sulphur, two, electronegative
Chemical Bonding
8 Likes
Answer
Sulphur, two, electronegative
Reason — X has electronic configuration 2, 8, 6. The molecule has total 16 electrons, So, the atomic number is 16. Hence, the element X is Sulphur (S). To achieve the nearest nobel gas configuration of argon(2,8,8), sulphur will gain two electrons. So, Y is 2. Gaining of electrons forms a negative ion. Thus, sulphur will be electronegative.
Answered By
3 Likes
Related Questions
When electrolysis of molten lead bromide is carried out, the products formed at the respective electrodes are:
At the positive electrons At the negative electrode 1. Bromine Lead 2. Bromine Hydrogen 3. Lead Bromine 4. Lead Oxygen The following are the structural diagrams of certain hydrocarbons:
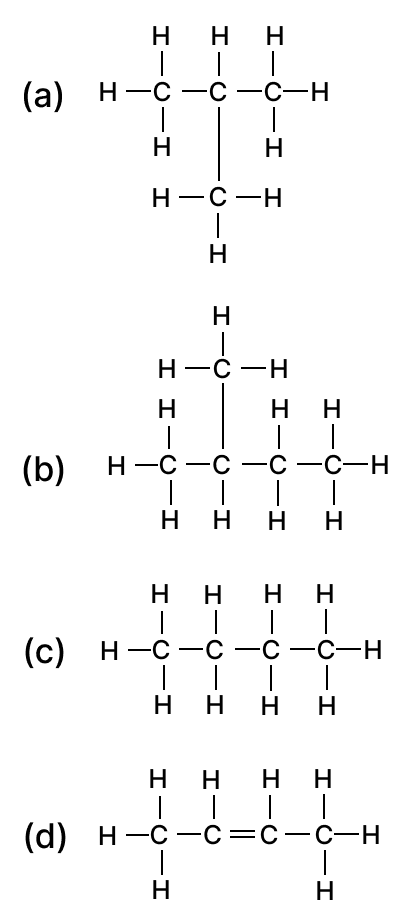
Which two structures are related to each other?
- A and B
- B and C
- C and D
- A and C
Which of the following arrangements is INCORRECT as per the property stated against it?
- Li > Be > N > O (Metallic character)
- CI > F > Br > I (Electron gain enthalpy)
- O2- > F - > Mg2+ > Na+(Ionic radii)
- I > Br > CI > F (Number of shells)
Baking soda (NaHCO3), when added to vinegar, evolves a gas. Which of these statements is true about the evolution of gas?
I. It turns limewater milky.
II. It extinguishes the burning splinter.
III. It acts as a non-metallic oxide
IV. It has a pungent odour.- I and IV
- I and II
- I, II and III
- III and IV