Physics
In an experiment to measure the temperature of the flame of a Bunsen burner, a lump of copper of mass 0.12 kg is heated on the flame for a long time. The copper then is quickly transferred into a beaker of negligible heat capacity containing 0.84 kg of water and the temperature of the water rose from 15 °C to 35 °C. Calculate the temperature of the flame.
[Given : Specific heat capacity of copper = 0.4 J g-1 °C-1, Specific heat capacity of water = 4.2 J g-1 °C-1]
Related Questions
An object is placed at a distance 24 cm in front of a convex lens of focal length 8 cm.
(a) What is the nature of the image so formed?
(b) Calculate the distance of the image from the lens.
The diagram below shows a cooling curve for a substance X :

(a) State the temperatures at which the substance condenses.
(b) Mention the temperature range in which the substance is in its liquid state.
(c) State True or False: The amount of heat released when a substance is cooled by 10°C in its liquid state is greater than the heat released when it is cooled by the same amount in its solid state.
A metal rod AB of length 80 cm is balanced at 45 cm from the end A with 100 gf weights suspended from the two ends.
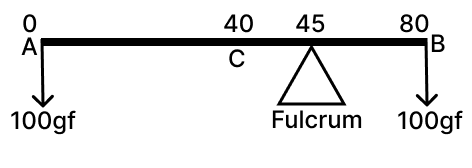
(a) If this rod is cut at the centre C, then compare the weight of AC to the weight of BC.
(b) Give a reason for your answer in (a)
For each of the following scenarios, state whether the work done by gravity is positive, negative, or zero.
(a) a person walks on a levelled road.
(b) a person climbs a ladder.
(c) a car in neutral gear is coming down the slope.