Chemistry
FeS + H2SO4 ⟶ FeSO4 + H2S is an example of:
- Displacement reaction
- Double decomposition reaction
- Decomposition reaction
- Combination reaction
Answer
Double decomposition reaction
Reason — A chemical reaction in which both reactants [compounds] are decomposed to give two new compounds by exchanging their radicals is called a Double decomposition or double displacement reaction.
AB + CD ⟶ AD + CB
No single element is replacing another element, so it can't be displacement reaction. A single compound breaking into simpler substances is not happening, so it can't be decomposition reaction. Two reactants combining into one product is not the case here, so it can't be combination reaction.
Related Questions
Which one of these is a chemical change?
- Water changes to steam
- Dissolution of sugar in water
- Combustion of LPG
- Liquefying ammonia
The reaction of neutralization is a :
- Displacement reaction
- Double decomposition
- Combination reaction
- Combination reaction between two compounds
Which of the following statements is/are correct with respect to the figure shown alongside?
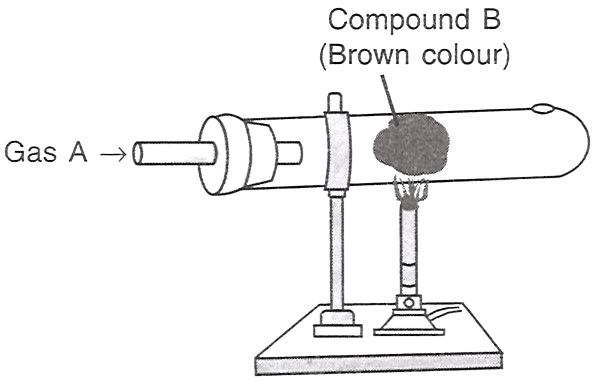
P — A is HCl, B is iron.
Q — A is Cl2, B is copper.
R — A is Cl2, B is iron.
Only P
Only Q
Only R
Both P and R
Aman performed the following four experiments
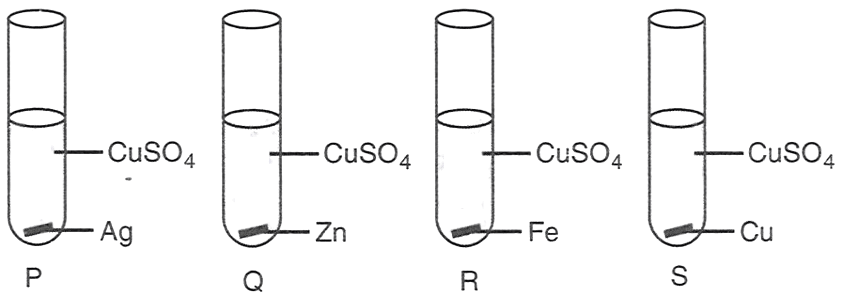
The experiments in which the blue colour of the solution will fade are :
P and Q
P, Q and R
Q and R
Q, R and S