Science
The formation of magnesium oxide is correctly shown in option :
options
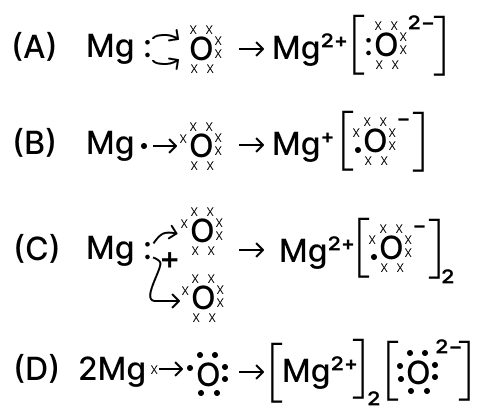
Metals & Non-Metals
1 Like
Answer
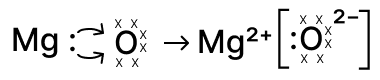
Reason — When magnesium reacts with oxygen each Mg atom loses its two valence electrons to form Mg2+ and an oxygen atom (which has six valence electrons) gains those two electrons to complete its octet and form O2-.
The correct electron-dot representation shows both valence electrons of Mg being transferred to oxygen, leaving Mg2+ (no valence dots) and the oxide ion O2- with eight electrons as shown below :
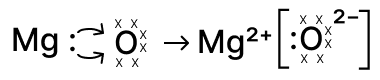
Answered By
1 Like
Related Questions
Reaction between two elements A and B, forms a compound C. A loses electrons and B gains electrons. Which one of the following properties will not be shown by compound C?
options
- It has high melting point.
- It is highly soluble in water.
- It has weak electrostatic forces of attraction between its oppositely charged ions.
- It conducts electricity in its molten state or aqueous solution
The metals obtained from their molten chlorides by the process of electrolytic reduction are :
options
- Gold and silver
- Calcium and magnesium
- Aluminium and silver
- Sodium and iron
Secretion of less saliva in mouth will effect the conversion of :
options
- Proteins into amino acids
- Fats into fatty acids and glycerol
- Starch into simple sugars
- Sugars into alcohol
The plant hormone whose concentration stimulates the cells to grow longer on the side of the shoot which is away from light is :
options
- Cytokinins
- Gibberellins
- Adrenaline
- Auxins