Chemistry
K, Na, Ca, Mg, Al, Zn, Fe, Pb, Cu, H, Hg, Ag
Pick an element from the reactivity series given above and write the equations for the the following:
(a) Metal hydroxide on heating forms metal oxide and water vapour.
(b) Metal hydroxide on heating forms metal, oxygen and water vapours.
(c) Metal nitrate decomposes to give two products only.
(d) Metal nitrate on heating forms metal oxide, nitrogen dioxide and oxygen.
(e) Metal nitrate on heating forms metal, nitrogen dioxide and oxygen.
(f) Metal carbonate which is stable to heat.
(g) Metal carbonate which forms metal oxide and carbon dioxide on heating.
(h) The heating effect on bivalent metal hydrogen carbonate.
Related Questions
Take sodium sulphate solution in a test tube and add barium chloride solution to it dropwise.
(a) What do you observe?
(b) Why is the reaction fast?
(c) Which type of reaction is it?
NaCl + AgNO3 ⟶ AgCl + NaNO3
(a) What will you observe in the above chemical change?
(b) Why is this reaction known as a double displacement reaction?
(c) Which reagent can be used to test the chloride radical?
KI + Cl2 ⟶ KCl + I2
(a) Balance the equation given above.
(b) Which type of reaction is it ?
(c) Which element is more reactive ?
(d) What will be the product formed if KCl reacts with I2 ?
See the diagram given below and answer the following questions:
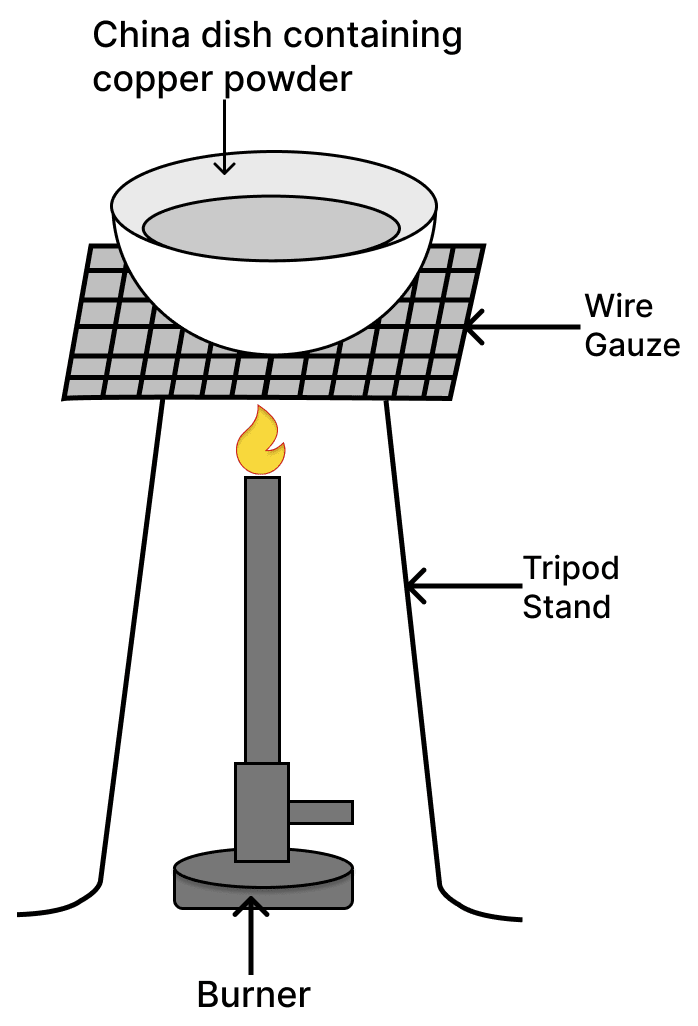
(a) Write the chemical reaction involved in the process.
(b) Mention the colour of :
(i) copper powder and
(ii) the substance formed after heating it.(c) Can we reverse the above reaction ? If yes, write the equation for the reverse reaction and state the substance that undergoes oxidation and the substance that undergoes reduction.