Chemistry
See the diagram given below and answer the following questions:
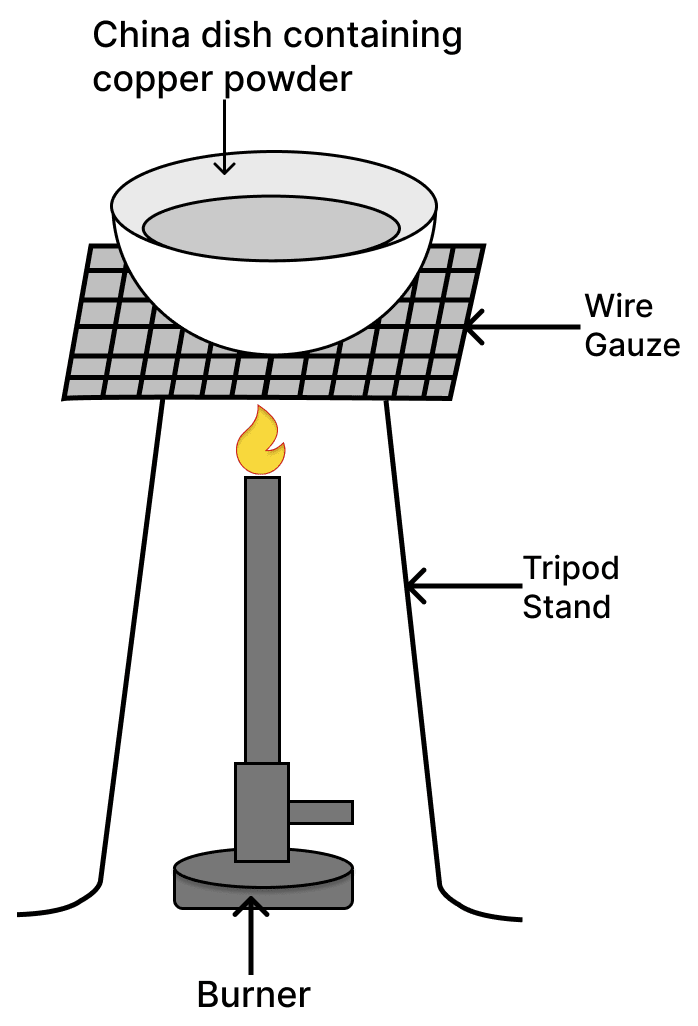
(a) Write the chemical reaction involved in the process.
(b) Mention the colour of :
(i) copper powder and
(ii) the substance formed after heating it.
(c) Can we reverse the above reaction ? If yes, write the equation for the reverse reaction and state the substance that undergoes oxidation and the substance that undergoes reduction.
Answer
(a) 2Cu + O2 ⟶ 2CuO
(b)
(i) The colour of copper powder is reddish brown.
(ii) The substance formed after heating, copper(II) oxide, is black in colour.
(c) Yes, the reaction can be reversed by reducing copper(II) oxide with hydrogen.
CuO + H2 ⟶ Cu + H2O
In this reaction, hydrogen is oxidised to water, and copper(II) oxide is reduced to copper.
Related Questions
K, Na, Ca, Mg, Al, Zn, Fe, Pb, Cu, H, Hg, Ag
Pick an element from the reactivity series given above and write the equations for the the following:
(a) Metal hydroxide on heating forms metal oxide and water vapour.
(b) Metal hydroxide on heating forms metal, oxygen and water vapours.
(c) Metal nitrate decomposes to give two products only.
(d) Metal nitrate on heating forms metal oxide, nitrogen dioxide and oxygen.
(e) Metal nitrate on heating forms metal, nitrogen dioxide and oxygen.
(f) Metal carbonate which is stable to heat.
(g) Metal carbonate which forms metal oxide and carbon dioxide on heating.
(h) The heating effect on bivalent metal hydrogen carbonate.
KI + Cl2 ⟶ KCl + I2
(a) Balance the equation given above.
(b) Which type of reaction is it ?
(c) Which element is more reactive ?
(d) What will be the product formed if KCl reacts with I2 ?
Assertion (A): Zinc can displace copper from aqueous copper sulphate solution.
Reason (R): Copper is placed above zinc in the reactivity series.
- Both A and R are true and R is the correct explanation of A.
- Both A and R are true but R is not the correct explanation of A.
- A is true but R is false.
- A is false but R is true.
Assertion (A): In a redox reaction, the electron gaining species acts as a reducing agent.
Reason (R): Oxidation and reduction reactions occur simultaneously in redox reaction.
- Both A and R are true and R is the correct explanation of A.
- Both A and R are true but R is not the correct explanation of A.
- A is true but R is false.
- A is false but R is true.