Which of the following metals does not react with water or acid to produce hydrogen gas?
- Magnesium
- Iron
- Copper
- Zinc
Answer
Copper
Reason — Copper (Cu) is less reactive and lies below hydrogen in the reactivity series, so it cannot displace hydrogen from dilute acids and therefore does not produce hydrogen gas. Also, copper does not react with water (cold or hot) to give hydrogen.
Magnesium reacts with hot water and acids to produce hydrogen gas. Iron reacts slowly with water (steam) and with acids to release hydrogen.
Which of the following compounds is formed along with hydrogen gas when magnesium reacts with steam ?
- Mg(OH)2
- MgO
- MgH2
- MgCl2
Answer
MgO
Reason — When magnesium reacts with steam (water vapour), it forms magnesium oxide (MgO) along with hydrogen gas (H2).
Mg(s) + H2O(g) ⟶ MgO(s) + H2(g)
Mg(OH)2 is formed when magnesium reacts with cold water, not steam. MgH2 is not formed in this reaction. MgCl2 is formed when magnesium reacts with hydrochloric acid, not steam.
The gas produced at anode when electrolysis of water is done is :
- H2
- O2
- SO2
- None
Answer
O2
Reason — During water electrolysis, hydrogen gas (H2) is produced at the cathode (negative electrode), and oxygen gas (O2) is produced at the anode (positive electrode).
Equal volumes of hydrogen and chlorine are exposed to diffused sunlight to prepare
- hydrogen chloride
- water
- sodium hydroxide
- hydrochloric acid
Answer
hydrogen chloride
Reason — In the presence of diffused sunlight, the hydrogen and chlorine molecules undergo a combination reaction to form hydrogen chloride.
H2(g) + Cl2(g) 2HCl(g)
The metal which reacts with cold water to produce hydrogen is
- magnesium
- aluminium
- calcium
- iron
Answer
calcium
Reason — Calcium (Ca) reacts readily with cold water to form calcium hydroxide (Ca(OH)2) and hydrogen gas (H2).
Ca(s) + 2H2O(l) ⟶ Ca(OH)2(aq) + H2(g)
Magnesium reacts very slowly with cold water, but reacts readily with hot water/steam. Aluminium does not react with cold water due to a protective oxide layer. Iron reacts only with steam, not with cold water.
In metal reactivity series, the more reactive metals are at
- top
- bottom
- middle
- none of these
Answer
top
Reason — The reactivity series is a list of metals arranged in order of their ability to displace other metals from solutions or react with substances like water and acids. The most reactive metals, such as potassium and sodium, are at the top, while less reactive metals like gold and platinum are at the bottom.
Hydrogen is responsible for producing
- heat and light
- hydrogenated oil
- fertilizers
- all of the above
Answer
all of the above
Reason — Hydrogen is used as a fuel to produce heat and light, in the hydrogenation of vegetable oils to produce hydrogenated oil, and in the manufacture of ammonia used for fertilisers.
Hydrogen is
- combustible
- non-combustible
- supporter of combustion
- neither supporter nor combustible
Answer
combustible
Reason — Hydrogen is combustible in nature, but is itself a non-supporter of combustion.
Water gas is a mixture of
- carbon monoxide and oxygen
- carbon monoxide and hydrogen
- hydrogen and oxygen
- hydrogen and nitrogen.
Answer
carbon monoxide and hydrogen
Reason — Water gas is a mixture of carbon monoxide (CO) and hydrogen (H2).
Assertion (A): On the earth, hydrogen is mostly found in the form of compounds.
Reason (R): Hydrogen is a highly reactive element.
- Both A and R are true and R is the correct explanation of A.
- Both A and R are true but R is not the correct explanation of A.
- A is true but R is false.
- A is false but R is true.
Answer
Both A and R are true and R is the correct explanation of A.
Explanation — A is true as hydrogen readily reacts with other elements, especially oxygen, to form compounds like water, and with metals as well.
R is true as this reactivity is one of the reasons it is mostly found in compounds.
Therefore, Both A and R are true and R is the correct explanation of A is the correct option.
Assertion (A): In the laboratory preparation of hydrogen, granulated zinc is reacted with dilute acid.
Reason (R): Granulated zinc contains copper as an impurity.
- Both A and R are true and R is the correct explanation of A.
- Both A and R are true but R is not the correct explanation of A.
- A is true but R is false.
- A is false but R is true.
Answer
Both A and R are true but R is not the correct explanation of A.
Explanation — In the laboratory, hydrogen is commonly prepared by reacting granulated zinc with a dilute acid (dil. HCl / dil. H2SO4). So, Assertion (A) is true.
Granulated zinc may contain small impurities such as copper, which can help the reaction start faster. So, Reason (R) is true.
However, Reason (R) is not the correct explanation of Assertion (A) as granulated zinc is mainly used because its rough, uneven surface provides larger effective surface area, so hydrogen is evolved at a steady and controllable rate.
Therefore, Both A and R are true but R is not the correct explanation of A is the correct option.
Assertion (A): Pure hydrogen burns with a pop sound in air.
Reason (R): Hydrogen forms an explosive mixture with air.
- Both A and R are true and R is the correct explanation of A.
- Both A and R are true but R is not the correct explanation of A.
- A is true but R is false.
- A is false but R is true.
Answer
A is false but R is true.
Explanation — Pure hydrogen burns quietly (pale blue flame). The pop sound is heard only when hydrogen is mixed with air/oxygen. So, Assertion (A) is false.
Hydrogen forms an explosive mixture with air, which produces the pop on ignition. So, Reason (R) is true.
Therefore, A is false but R is true is the correct option.
Assertion (A): Hydrogen is used for the extraction of less reactive metals like copper and lead.
Reason (R): Hydrogen is a good reducing agent.
- Both A and R are true and R is the correct explanation of A.
- Both A and R are true but R is not the correct explanation of A.
- A is true but R is false.
- A is false but R is true.
Answer
Both A and R are true and R is the correct explanation of A.
Explanation — A is true, when hydrogen gas is passed over hot metallic oxides of copper, lead, iron, etc., it removes oxygen from them and thus reduces them to their corresponding metals.
R is true as hydrogen acts as a good reducing agent.
Therefore, Both A and R are true and R is the correct explanation of A is the correct option.
Assertion (A): Oxyhydrogen flame is used for cutting and welding metals.
Reason (R): For cutting and welding metals, a low temperature is required.
- Both A and R are true and R is the correct explanation of A.
- Both A and R are true but R is not the correct explanation of A.
- A is true but R is false.
- A is false but R is true.
Answer
A is true but R is false.
Explanation — Oxyhydrogen flame is used for cutting and welding metals. So, Assertion (A) is true.
For cutting and welding metals, a high temperature is required. Oxyhydrogen flame has a temperature of about 2800°C–3000°C, which is sufficient to melt the metals. So, Reason (R) is false.
Therefore, A is true but R is false is the correct option.
Fill in the blanks:
(a) Hydrogen is ............... than air.
(b) Hydrogen is ............... soluble in water.
(c) Hydrogen burns with a ............... flame and ............... sound is heard.
(d) A metal ............... hydrogen in the reactivity series gives hydrogen with ............... .
(e) Hydrogen reacts with metal oxides to form ............... and ............... .
(f) Oxidation is the removal of ............... or addition of ............... .
(g) In redox reactions, oxidation and reduction occur ............... .
Answer
(a) lighter
(b) sparingly
(c) pale blue, pop
(d) above, dilute acids
(e) metal, water
(f) hydrogen, oxygen
(g) simultaneously
Indicate which of the following statements are true and which are false and correct the false statements:
(a) Hydrogen molecule is monovalent.
(b) The removal of hydrogen from a substance is called reduction.
(c) Nitric acid cannot be used to prepare hydrogen by its action on active metals.
(d) The reaction between hydrogen and nitrogen to form ammonia is reversible.
(e) Zinc can liberate hydrogen from water, acid and alkali solution.
(f) Hydrogen is combustible as well as a supporter of combustion.
(g) Hydrogen gas is easily liquefiable.
Answer
(a) False
Corrected statement — Hydrogen atom is monovalent; hydrogen molecule is diatomic.
(b) False
Corrected statement — The removal of hydrogen from a substance is called oxidation.
(c) True
(d) True
(e) True
(f) False
Corrected statement — Hydrogen is combustible but it is a non-supporter of combustion.
(g) False
Corrected statement — Hydrogen gas cannot be easily liquefied.
Match the statements in Column A with the words in Column B.
| Column A | Column B |
|---|---|
| (a) A metal which reacts with cold water to form hydrogen. | (i) Reduction |
| (b) A gas which is inflammable and a non-supporter of combustion. | (ii) Hydrogenation |
| (c) A process in which vanaspati ghee is prepared from vegetable oils. | (iii) Oxidation |
| (d) The removal of hydrogen or addition of oxygen. | (iv) Sodium |
| (e) The addition of hydrogen or removal of oxygen. | (v) Hydrogen |
Answer
| Column A | Column B |
|---|---|
| (a) A metal which reacts with cold water to form hydrogen. | (iv) Sodium |
| (b) A gas which is inflammable and a non-supporter of combustion. | (v) Hydrogen |
| (c) A process in which vanaspati ghee is prepared from vegetable oils. | (ii) Hydrogenation |
| (d) The removal of hydrogen or addition of oxygen. | (iii) Oxidation |
| (e) The addition of hydrogen or removal of oxygen. | (i) Reduction |
Name the following:
(a) The process in which oxygen is added or hydrogen is removed.
(b) A process by which hydrogen gas is manufactured.
(c) A solid conductor through which current enters or leaves the electrolyte.
(d) The charged particles present in an electrolyte.
(e) A substance which removes oxygen from a compound.
Answer
(a) Oxidation
(b) Bosch process
(c) Electrode
(d) Ions
(e) Reducing agent
Give examples :
(a) Two metals which give hydrogen with cold water.
(b) A metal which liberates hydrogen only when steam is passed over red hot metal.
(c) A metallic oxide which can be reduced into metal by hydrogen.
(d) An electrolyte.
(e) A metal of which an electrode is made.
Answer
(a) Sodium (Na) and Potassium (K)
(b) Iron (Fe)
(c) Copper(II) oxide (CuO)
(d) Copper(II) sulphate solution (CuSO4)
(e) Copper
Complete and balance the following chemical equations:
(a) H2 + ............... ⟶ 2HCl
(b) H2 + S ⟶ ...............
(c) Zn + ............... ⟶ ZnCl2 + H2
(d) CuO + ............... ⟶ Cu + ...............
(e) Fe + H2O ⟶ ............... + ...............
(f) K + H2O ⟶ ............... + ...............
Answer
(a) H2 + Cl2 ⟶ 2HCl
(b) H2 + S ⟶ H2S
(c) Zn + 2HCl ⟶ ZnCl2 + H2
(d) CuO + H2 ⟶ Cu + H2O
(e) 3Fe + 4H2O (steam) ⟶ Fe3O4 + 4H2
(f) 2K + 2H2O ⟶ 2KOH + H2
Define:
(a) catalytic hydrogenation
(b) oxidation
(c) reduction
(d) redox reaction
(e) electrolysis
(f) electrolyte
Answer
(a) Catalytic hydrogenation is a process by which hydrogen gas is passed through vegetable oils in the presence of a catalyst like Ni, Pt or Pd to convert the oils into vanaspati ghee.
(b) Oxidation is a chemical process that involves addition of oxygen to a substance or removal of hydrogen from a substance.
(c) Reduction is a chemical process that involves addition of hydrogen to a substance or removal of oxygen from a substance.
(d) Redox reactions are those in which reduction and oxidation both take place simultaneously i.e., one substance is reduced while the other gets oxidised.
(e) Electrolysis is a process in which an electric current is passed through an aqueous solution or a molten state of a compound to bring about a chemical change.
(f) An electrolyte is a compound which, in aqueous solution or molten state, conducts electricity due to the presence of free and mobile ions.
How would you show that hydrogen gas is neutral in nature ?
Answer
Hydrogen gas is neutral to litmus, i.e. no change is observed in the colour of either moist blue or red litmus paper when it is introduced into a jar containing hydrogen. This proves that hydrogen gas is neutral in nature.
Give reasons for the following:
Hydrogen can be used as a fuel.
Answer
(a) Hydrogen has high heat of combustion and is pollution free so it is used as a fuel. Liquid hydrogen is used as a rocket fuel.
Give reasons for the following:
Though hydrogen is lighter than air it cannot be collected by downward displacement of air.
Answer
Though hydrogen is lighter than air it cannot be collected by downward displacement of air because a mixture of hydrogen and air can lead to an explosion so it is not safe to collect hydrogen gas by this method.
Give reasons for the following:
A pop sound is produced when hydrogen gas is burnt.
Answer
A pop sound is produced when hydrogen gas is mixed with air/oxygen and burnt because hydrogen forms an explosive mixture with air/oxygen, which burns suddenly on ignition.
Give reasons for the following:
Helium has replaced hydrogen in weather forecasting balloons.
Answer
Hydrogen gas is highly inflammable in nature, so it is replaced by helium for filling in weather forecasting balloons.
Give reasons for the following:
Nitric acid is not used for the preparation of hydrogen gas.
Answer
Nitric acid is a strong oxidising agent (even when it is dilute). It oxidises hydrogen gas converting it into water which defeats the purpose. Hence, nitric acid is not used for the preparation of hydrogen gas.
The chemical equation given below represents a redox reaction.
PbO (Lead monoxide) + H2 ⟶ Pb (Lead) + H2O
(a) Name the reactant which gets :
(i) reduced
(ii) oxidised.
(b) Which of the reactants acts as an oxidising agent ?
Answer
(a)
(i) The reactant which gets reduced is PbO (Lead oxide).
(ii) The reactant which gets oxidised is H2 (Hydrogen gas).
(b) PbO (Lead monoxide) acts as the oxidising agent.
(a) Name a process by which hydrogen gas is manufactured.
(b) Give equations for the reactions.
(c) How is hydrogen separated from carbon dioxide and carbon monoxide?
Answer
(a) Bosch process
(b) Equations for the reactions of Bosch process are:
Production of water gas:
Reduction of steam by water gas:
(c) Hydrogen is separated from carbon dioxide by passing the mixture through water under pressure, where carbon dioxide gets dissolved leaving behind hydrogen. Carbon dioxide can also be separated by passing it through caustic potash (KOH) solution.
2KOH + CO2 ⟶ K2CO3 + H2O
Carbon monoxide is separated by passing through ammoniacal cuprous chloride in which carbon monoxide dissolves leaving behind hydrogen.
CuCl + CO + 2H2O ⟶ CuCl.CO.2H2O
Hydrogen is a good reducing agent. What do you understand by the above statement? Explain with the help of copper oxide as an example.
Answer
When hydrogen is passed over hot metallic oxides of copper, lead, iron, etc. it removes oxygen from them and thus reduces them to their corresponding metals. Hence, hydrogen acts as a good reducing agent.
When hydrogen gas is passed over heated copper oxide, copper oxide gets reduced to metallic copper by the removal of oxygen from it. The equation for the reaction is given below:
CuO + H2 ⟶ Cu + H2O
State four uses of hydrogen.
Answer
The four uses of hydrogen are:
- The oxy-hydrogen flame obtained by burning hydrogen and oxygen together has a very high temperature of about 2800°C–3000°C. Hence it is used for cutting and welding of metals.
- It is used as a fuel as hydrogen has high heat of combustion.
- It is used for hydrogenation of vegetable oil.
- It is used as a reducing agent in extraction of metals.
Give three differences between Cathode and anode.
Answer
| Cathode | Anode |
|---|---|
| The electrode connected to the negative terminal of the battery is called cathode. | The electrode connected to the positive terminal of the battery is called anode. |
| Current leaves the electrolyte through the cathode. | Current enters the electrolyte through the anode. |
| Cations migrate and discharge at the cathode. | Anions migrate and discharge at the anode. |
Give three differences between Cation and anion.
Answer
| Cation | Anion |
|---|---|
| Cations are positively charged atoms or group of atoms. | Anions are negatively charged atoms or group of atoms. |
| During electrolysis, cations migrate towards the cathode. | During electrolysis, anions migrate towards the anode. |
| Examples : Na+, Ba2+, Ca2+, etc. | Examples : Cl-, OH-, Br-, etc. |
(a) Name the chemicals required to prepare hydrogen gas in the laboratory.
(b) Give a balanced chemical equation for the reaction.
(c) Draw a neat and well-labelled diagram for the laboratory preparation of hydrogen.
(d) How is hydrogen gas collected? Why?
(e) Give one chemical test for hydrogen.
Answer
(a) Granulated zinc and dilute hydrochloric acid are the chemicals required to prepare hydrogen gas in the laboratory.
(b) Zn + 2HCl ⟶ ZnCl2 + H2 (g)
(c) The diagram below shows the laboratory preparation of hydrogen gas:

(d) Hydrogen gas is collected by downward displacement of water as it is sparingly soluble in water. Though hydrogen gas is lighter than air, it is not collected by downward displacement of air as a mixture of hydrogen and air can lead to an explosion.
(e) Hydrogen gas burns in air with a pop sound. This is because of the presence of impurities in it. This method is widely used as a test to identify hydrogen.
The diagram given below shows electrolysis of water. Answer the following questions :
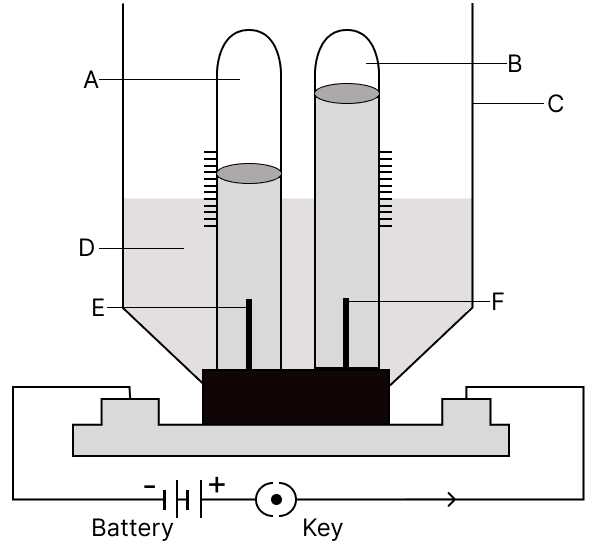
(a) Label A, B, C, D, E and F.
(b) Why is acidulated water used ?
(c) What is the ratio by volume for the gases collected ?
Answer
(a)
A → Hydrogen (H2)
B → Oxygen (O2)
C → Electrolytic Cell
D → Acidulated Water (Electrolyte)
E → Cathode
F → Anode
(b) Acidulated water is used as pure water is a bad conductor of electricity. When a small amount of acid, alkali or salt is added to it, only then it conducts electricity.
(c) Ratio by volume of the gases collected
Hydrogen : Oxygen = 2 : 1
Hydrogen is the most abundant element in the universe. On earth, however, it is rarely found in the free state because of its high reactivity, and usually occurs in combined forms such as water and hydrocarbons. Owing to its unique properties, hydrogen has a wide range of applications. It is used in cutting and welding metals, as a reducing agent for extracting metals from their oxides, in the preparation of vanaspati ghee from vegetable oils and in the manufacture of many important chemicals. Today, hydrogen is widely regarded as a potential clean fuel of the future.
(a) Why is hydrogen known as a clean fuel ?
(b) Define the process used to prepare vanaspati ghee from vegetable oils. Name a catalyst used in this process.
(c) Why is hydrogen used for cutting and welding metals ?
(d) Why is hydrogen not used in weather forecasting balloons anymore ?
(e) Name the gas which has replaced hydrogen in these balloons. Why ?
Answer
(a) Hydrogen is called a clean fuel because on burning it produces only water. It does not produce smoke, carbon dioxide, or harmful gases, so it does not pollute the environment.
(b) The process used to prepare vanaspati ghee from vegetable oils is called catalytic hydrogenation. In this process, hydrogen gas is passed through vegetable oil in the presence of a catalyst such as nickel to convert it into vanaspati ghee.
(c) Hydrogen is used for cutting and welding metals because it burns in oxygen to produce an oxy-hydrogen flame of very high temperature. This high heat is useful for cutting and welding metals.
(d) Hydrogen is not used in weather forecasting balloons anymore because it is highly inflammable and can catch fire easily. It may cause explosions, making it unsafe.
(e) Helium has replaced hydrogen in these balloons because it is non-flammable and lighter than air, so it can lift balloons safely.