One of the characteristics of a chemical reaction is:
- Catalyst
- Chemical bond
- Effervescence
- Evolution of gas
Answer
Evolution of gas
Reason — One of the characteristics of a chemical reaction is the evolution of a gas. This means that during some chemical reactions, one or more of the products can be a gas.
A catalyst is a substance that speeds up a reaction, not a characteristic of the reaction itself. Chemical bonds are part of substances, not a visible sign of a chemical reaction. Effervescence (bubbling) is one specific type of gas evolution, but not always present in all reactions.
When potassium chlorate is heated, the gas evolved is:
- Hydrogen
- Chlorine
- Oxygen
- Nitrogen
Answer
Oxygen
Reason — On being heated to 700°C, potassium chlorate decomposes to evolve oxygen gas.
A solution of silver nitrate is added to a solution of sodium chloride. The colour of the precipitate formed is:
- Black
- White
- Grey
- Green
Answer
White
Reason — When a solution of silver nitrate is added to a solution of sodium chloride, a white insoluble substance (precipitate), silver chloride, is formed.
AgNO3 (aq) + NaCl (aq) → AgCl↓ (white ppt.) + NaNO3 (aq)
Which of the following acts as a catalyst in the manufacture of ammonia gas from nitrogen and hydrogen?
- Manganese dioxide
- Platinum
- Iron
- Molybdenum
Answer
Iron
Reason — Finely divided iron is used as a positive catalyst in the manufacture of ammonia from hydrogen and nitrogen.
Manganese dioxide is used as a catalyst in decomposition reactions. Platinum is used in reactions like catalytic oxidation. Molybdenum is sometimes used as a promoter with iron.
The process of photosynthesis takes place in the presence of:
- Electricity
- Heat
- Light
- Sound
Answer
Light
Reason — Some chemical reactions like Photosynthesis can take place only in the presence of light. They are called photochemical reactions.
Electricity is not required for photosynthesis. Heat may affect the rate of photosynthesis, but it is not essential for the process. Sound has no role in photosynthesis.
Define :
(a) Chemical reaction
(b) Catalyst
(c) Promoter.
Answer
(a) Chemical reaction — Any chemical change in matter which involves transformation into one or more substances with entirely different properties is called a chemical reaction.
(b) Catalyst — A catalyst is a substance that either increases or decreases the rate of a chemical reaction without itself undergoing any chemical change during the reaction.
(c) Promoter — Substances that improve the efficiency of a catalyst are called promoters. For example, molybdenum acts as a promoter to increase the efficiency of the catalyst, iron, in the formation of ammonia gas from hydrogen and nitrogen.
What is a chemical bond?
Answer
A chemical bond is the attractive force that holds the atoms or ions of a molecule together in a compound.
What happens during a chemical reaction?
Answer
A chemical reaction involves breaking of chemical bonds between the atoms or groups of atoms of reacting substances and rearrangement of atoms making new bonds to form new substances with absorption or release of energy in the form of heat and light normally.
Differentiate between reactants and products.
Answer
Differences between Reactants and Products
| Reactants | Products |
|---|---|
| The substances that are used as the starting material and which react with one another are called reactants. | The substances which are formed as a result of the reaction are called products. |
| Reactants are written on the left hand side of the equation. | Products are written on the right hand side of the equation. |
| In equation C + O2 ⟶ CO2, C and O2 are reactants. | In equation C + O2 ⟶ CO2, CO2 is product. |
Name three biochemical catalysts (enzymes) found in human body.
Answer
The three biochemical catalysts found in the human body are amylase, trypsin, pepsin.
State two characteristics of chemical reactions.
Answer
Two characteristics of chemical reaction are given below:
Evolution of gas : In many chemical reactions, one of the products is a gas.
Example- When zinc reacts with dilute sulphuric acid, hydrogen gas is evolved with an effervescence.Change of colour : Certain chemical reactions are characterised by a change in the colour of the reactants.
Example- When a few pieces of iron are dropped into a blue coloured copper sulphate solution, the blue colour of the solution fades and eventually turns into light green.
Complete and balance the following chemical equations:
(a) N2 + O2 ⟶
(b) H2S + Cl2 ⟶
(c) Na + H2O ⟶
(d) NaCl + AgNO3 ⟶
(e) Zn + H2SO4 (dil.) ⟶
(f) FeSO4 (aq) + NaOH (aq) ⟶
(g) Pb(NO3)2
(h) BaCl2 (aq) + H2SO4 (aq) ⟶
Answer
(a) N2 + O2 2NO
(b) H2S + Cl2 ⟶ 2HCl ↑ + S
(c) 2Na + 2H2O ⟶ 2NaOH + H2 ↑
(d) NaCl + AgNO3 ⟶ NaNO3 + AgCl ↓
(e) Zn + H2SO4 (dil.) ⟶ ZnSO4 + H2 ↑
(f) FeSO4 (aq) + 2NaOH (aq) ⟶ Fe(OH)2 ↓ + Na2SO4 (aq)
(g) 2Pb(NO3)2 2PbO + 4NO2 ↑ + O2 ↑
(h) BaCl2 (aq) + H2SO4 (aq) ⟶ BaSO4 ↓ + 2HCl
Illustrate the following characteristics of a chemical reaction by giving a suitable example of each:
(a) evolution of a gas
(b) change of colour
(c) change in state
Answer
(a) In many chemical reactions, one of the products is a gas.
When zinc reacts with dilute sulphuric acid, hydrogen gas is evolved with an effervescence.
Zn + H2SO4 (dil.) ⟶ ZnSO4 + H2 (g)
(b) Certain chemical reactions are characterised by a change in the colour of the reactants.
When a few pieces of iron are dropped into a blue coloured copper sulphate solution, the blue colour of the solution fades and turns into light green.
Fe + CuSO4 (aq) ⟶ FeSO4 + Cu
(c) In many chemical reactions a change of state is observed.
Ammonia and hydrogen chloride gases react to produce a white solid of ammonium chloride.
NH3 (g) + HCl (g) ⟶ NH4Cl (s)
What are positive and negative catalysts? Give one example of each.
Answer
Positive catalyst : When a catalyst increases the rate of a chemical reaction, it is known as a positive catalyst.
Example — Finely divided iron is used as a positive catalyst in the manufacture of ammonia from hydrogen and nitrogen.
Negative catalyst : When a substance decreases the rate of a chemical reaction, it is known as a negative catalyst or inhibitor.
Example — Phosphoric acid acts as a negative catalyst to decrease the rate of decomposition of hydrogen peroxide.
What do you observe when:
(a) dilute sulphuric acid is added to granulated zinc?
(b) a few pieces of iron are dropped in a blue solution of copper sulphate?
(c) silver nitrate is added to a solution of sodium chloride?
(d) ferrous sulphate solution is added to an aqueous solution of sodium hydroxide?
(e) solid lead nitrate is heated?
(f) when dilute sulphuric acid is added to barium chloride solution?
Answer
(a) When dilute sulphuric acid is added to granulated zinc, hydrogen gas is evolved with an effervescence.
Zn + H2SO4 (dil.) ⟶ ZnSO4 + H2 (g)
(b) When a few pieces of iron are dropped into a blue coloured copper sulphate solution, the blue colour of the solution fades and turns into light green.
Fe + CuSO4 (aq) ⟶ FeSO4 + Cu
(c) When a solution of silver nitrate is added to a solution of sodium chloride, a white insoluble precipitate of silver chloride is formed.
AgNO3 (aq) + NaCl (aq) ⟶ NaNO3 (aq) + AgCl ↓
(d) When ferrous sulphate solution is added to sodium hydroxide solution, a dirty green precipitate of ferrous hydroxide is formed.
FeSO4 (aq) + 2NaOH (aq) ⟶ Fe(OH)2 ↓ + Na2SO4 (aq)
(e) When solid lead nitrate is heated strongly, it decomposes to produce light yellow solid lead monoxide, reddish brown nitrogen dioxide gas and colourless oxygen gas.
2Pb(NO3)2 ⟶ 2PbO + 4NO2 ↑ + O2 ↑
(f) When dilute sulphuric acid is added to barium chloride solution, a white precipitate of barium sulphate is formed.
BaCl2 + H2SO4 ⟶ BaSO4 ↓ + 2HCl
State the conditions required for the following reactions to occur :
(a) Decomposition of water into hydrogen and oxygen.
(b) Formation of iron sulphide from iron and sulphur.
(c) Reaction between zinc and dilute hydrochloric acid to produce hydrogen gas.
(d) Decomposition of potassium chlorate into potassium chloride and oxygen.
Answer
(a) Water decomposes into hydrogen and oxygen (gases) when electric current is passed through it.
(b) Iron powder and sulphur powder react to form a new substance, iron sulphide only when they are heated together.
(c) Zinc reacts with dilute hydrochloric acid at ordinary temperature to produce zinc chloride and hydrogen gas.
(d) Usually, potassium chlorate decomposes to evolve oxygen gas on being heated at 700°C. However, when manganese dioxide is mixed with potassium chlorate as a catalyst (in the ratio 1 : 4), the decomposition takes place at a much lower temperature, i.e. at about 300°C.
Burning of a metal in air represents a:
- Combination reaction
- Displacement reaction
- Decomposition reaction
- Double displacement reaction
Answer
Combination reaction
Reason — A combination reaction occurs when two or more substances combine to form a single product. When a metal burns in air, it reacts with oxygen to form a metal oxide.
A displacement reaction occurs when one element replaces another in a compound. A decomposition reaction occurs when a compound breaks down into simpler substances. A double displacement reaction occurs when there is an exchange of ions between two compounds.
Which of the following reactions is a neutralization reaction ?
- When zinc reacts with dilute HCl
- Reaction between barium chloride solution and dilute H2SO4
- When carbon burns in air
- Sodium hydroxide reacts with dilute H2SO4
Answer
Sodium hydroxide reacts with dilute H2SO4
Reason — A neutralisation reaction occurs when an acid reacts with a base to form water and salt. In this case, sodium hydroxide (NaOH) is a base, and dilute sulphuric acid (H2SO4) is an acid. When they react, they form water (H2O) and sodium sulphate (Na2SO4), which is a salt.
2NaOH(aq) + H2SO4(aq) → Na2SO4(aq) + 2H2O(l)
Zinc with dilute HCl is a displacement reaction. Barium chloride with dilute (H2SO4) is a double displacement reaction forming a precipitate. Carbon burning in air is a combination reaction.
Which of the following oxides produces salt upon reacting with sodium hydroxide?
- Na2O
- CaO
- NO
- ZnO
Answer
ZnO
Reason — Zinc oxide (ZnO) is an amphoteric oxide, meaning it can react both as an acid and as a base. When it reacts with sodium hydroxide (NaOH), a salt (zincate) is formed along with water, which makes it an acid-base reaction.
ZnO(s) + 2NaOH(aq) → Na2ZnO2(aq) + H2O(l)
Sodium oxide is a basic oxide. It does not react with sodium hydroxide. Calcium oxide is also a basic oxide. It does not react with bases. Nitric oxide is a neutral oxide. It does not react with sodium hydroxide.
The acidic gas produced when lead nitrate is heated is:
- PbO
- NO2
- MgO
- N2O
Answer
NO2
Reason — When lead nitrate (Pb(NO3)2) is heated, it undergoes decomposition to form lead(II) oxide (PbO), nitrogen dioxide (NO2), and oxygen (O2). Nitrogen dioxide (NO2) is a reddish brown acidic gas produced during this reaction.
2Pb(NO3)2(s) → 2PbO(s) + 4NO2(g) + O2(g)
The gas produced when iron metal reacts with dilute hydrochloric acid is:
- oxygen
- hydrogen
- chlorine
- water vapour
Answer
hydrogen
Reason — When iron (Fe) reacts with dilute hydrochloric acid (HCl), the reaction produces hydrogen gas (H2) and ferrous chloride (FeCl2).
Fe(s) + 2HCl(aq) → FeCl2(aq) + H2(g)
When copper metal is added to a solution of silver nitrate, the colour of the solution changes to blue. This shows:
- copper is less reactive than silver
- copper is more reactive than silver
- both are equally reactive
- none of the above
Answer
copper is more reactive than silver
Reason — When copper (Cu) is added to a solution of silver nitrate (AgNO3), a displacement reaction occurs. Copper displaces silver from silver nitrate because copper is more reactive than silver.
Cu (s) + 2AgNO3 (aq) → Cu(NO3)2 (aq) + 2Ag (s)
Which of the following equations represents an endothermic reaction?
- C + O2 CO2
- CaO + H2O → Ca(OH)2
- NaOH + HCl → NaCl + H2O
- CaCO3 CaO + CO2
Answer
CaCO3 CaO + CO2
Reason — An endothermic reaction is a type of reaction that absorbs heat from the surroundings. This means energy is required to break bonds in the reactants, and the products have more energy than the reactants. The decomposition of calcium carbonate (CaCO3) into calcium oxide (CaO) and carbon dioxide (CO2) requires heat to proceed.
Assertion (A): A chemical reaction is a process which involves the transformation of one or more substances into new substances with entirely different properties.
Reason (R): Breaking of chemical bonds between the atoms of reacting substances takes place to form new bonds between the atoms forming new substances called products.
- Both A and R are true and R is the correct explanation of A.
- Both A and R are true but R is not the correct explanation of A.
- A is true but R is false.
- A is false but R is true.
Answer
Both A and R are true and R is the correct explanation of A.
Explanation — A is true as a chemical reaction involves the transformation of reactants into products with different properties.
R is true as this change occurs because the chemical bonds between the atoms in the reactants are broken, and new bonds are formed to create the products.
Therefore, Both A and R are true and R is the correct explanation of A is the correct option.
Assertion (A): A chemical equation must be balanced in order to follow the law of conservation of mass.
Reason (R): A chemical equation called as skeletal equation may not have equal number of atom of each element present in the reactants and products.
- Both A and R are true and R is the correct explanation of A.
- Both A and R are true but R is not the correct explanation of A.
- A is true but R is false.
- A is false but R is true.
Answer
Both A and R are true and R is the correct explanation of A.
Explanation — A is true. According to the law of conservation of mass, the total mass of reactants equals the total mass of products. Therefore, the number of atoms of each element must be the same on both sides of a chemical equation, so the equation must be balanced.
R is true as a skeletal equation is an unbalanced equation and may have unequal atoms of elements in reactants and products. Hence, balancing the equation is necessary, and R correctly explains A.
Therefore, Both A and R are true and R is the correct explanation of A is the correct option.
Assertion (A): The reaction of an acid with a base to produce salt and water is called a neutralisation reaction.
Reason (R): Neutralisation reaction is a double displacement reaction.
- Both A and R are true and R is the correct explanation of A.
- Both A and R are true but R is not the correct explanation of A.
- A is true but R is false.
- A is false but R is true.
Answer
Both A and R are true but R is not the correct explanation of A.
Explanation — A is true as the reaction of an acid with a base to form salt and water is called a neutralisation reaction.
R is true as this type of reaction is indeed a double displacement reaction, as the hydrogen ions (H⁺) from the acid and the hydroxide ions (OH⁻) from the base combine to form water, while the other ions combine to form the salt. However, these are two separate facts.
Therefore, Both A and R are true but R is not the correct explanation of A is the correct option.
Assertion (A): When a copper coil is dipped in an aqueous solution of silver nitrate, the colour of the solution slowly changes from colourless to blue.
Reason (R): Silver metal is more reactive than copper.
- Both A and R are true and R is the correct explanation of A.
- Both A and R are true but R is not the correct explanation of A.
- A is true but R is false.
- A is false but R is true.
Answer
A is true but R is false.
Explanation — A is true as when copper is placed in silver nitrate solution, solution turns blue due to formation of copper nitrate.
R is false since copper is chemically more reactive than silver, it displaces silver in the aqueous solution of silver nitrate to form copper nitrate.
Cu (s) + 2AgNO3(aq) → Cu(NO3)2(aq) + 2Ag (s)
Therefore, A is true but R is false is the correct option.
Assertion (A): When energy is absorbed during a chemical reaction, it is said to be an exothermic reaction.
Reason (R): A rise in temperature takes place during an exothermic reaction.
- Both A and R are true and R is the correct explanation of A.
- Both A and R are true but R is not the correct explanation of A.
- A is true but R is false.
- A is false but R is true.
Answer
A is false but R is true.
Explanation — Endothermic reactions absorb heat from the surroundings, while exothermic reactions release heat to the surroundings. Hence, if energy is absorbed, the reaction is endothermic, so Assertion (A) is false. In an exothermic reaction, heat released causes a rise in temperature, so Reason (R) is true.
Therefore, A is false but R is true is the correct option.
Fill in the blanks:
(a) A reaction in which two or more substances combine to form a single substance is called a ............... reaction.
(b) A ............... is a substance which changes the rate of a chemical reaction without undergoing a chemical change itself.
(c) The formation of gas bubbles in a liquid during a reaction is called ............... .
(d) The reaction between an acid and a base is called ............... .
(e) Soluble bases are called ............... .
(f) The chemical change involving iron and hydrochloric acid illustrates a ............... reaction.
(g) In the type of reaction called ............... two compounds exchange their positive and negative radicals respectively.
(h) A catalyst either ............... or ............... the rate of a chemical change but itself remains ............... at the end of the reaction.
(i) The chemical reaction between hydrogen and chlorine is a ............... reaction.
(j) When a piece of copper is added to silver nitrate solution, it turns ............... in colour.
Answer
(a) combination
(b) catalyst
(c) effervescence
(d) neutralisation reaction
(e) alkalis
(f) displacement
(g) double displacement
(h) increases, decreases, unchanged
(i) combination
(j) blue
Write true or false for the following and correct the false statements.
(a) When iron nails are put into a solution of copper sulphate, the solution becomes colourless after some time.
(b) Burning of fuel in air produces a large amount of energy in the form of heat and light.
(c) Exchange of radicals takes place when calcium oxide reacts with water.
(d) A neutral oxide does not change the colour of an indicator.
(e) A white solid is produced when lead nitrate is heated.
Answer
(a) False
Correct Statement — When iron nails are put into a solution of copper sulphate, the solution becomes light green after some time.
(b) True
(c) False
Correct Statement — Calcium oxide reacts with water to form calcium hydroxide; it is a combination reaction, not an exchange (double displacement) reaction.
(d) True
(e) False
Correct Statement — A pale yellow solid, reddish brown gas and colourless oxygen gas are produced when lead nitrate is heated.
Match the following :
| Column A | Column B |
|---|---|
| (a) It changes the rate of a chemical reaction | (i) Photosynthesis |
| (b) Attractive force between the atoms to form molecules | (ii) Amphoteric oxide |
| (c) Oxides of non-metals | (iii) Catalyst |
| (d) Lead monoxide | (iv) Chemical bond |
| (e) Light energy | (v) Acidic oxides |
Answer
| Column A | Column B |
|---|---|
| (a) It changes the rate of a chemical reaction | (iii) Catalyst |
| (b) Attractive force between the atoms to form molecules | (iv) Chemical bond |
| (c) Oxides of non-metals | (v) Acidic oxides |
| (d) Lead monoxide | (ii) Amphoteric oxide |
| (e) Light energy | (i) Photosynthesis |
Name the following :
(a) The substances formed due to a chemical reaction.
(b) An insoluble solid produced when two substances in solution form are mixed together.
(c) The compounds formed by the reaction between oxygen and any other element.
(d) The chemical reactions in which energy is released.
(e) A chemical reaction in which a substance breaks to give two or more simpler substances.
Answer
(a) Products
(b) Precipitate
(c) Oxides
(d) Exothermic reactions
(e) Decomposition reaction
Give one example for each of the following :
(a) An oxide which gives salts both with an acid and a base.
(b) An oxide which does not change the colour of indicators.
(c) A neutral oxide which is a universal solvent.
(d) An oxide which produces salt and water with an acid.
(e) A soluble metallic oxide which produces an alkali.
Answer
(a) Zinc oxide (ZnO)
(b) Carbon monoxide (CO)
(c) Water (H2O)
(d) Calcium oxide (CaO)
(e) Sodium oxide (Na2O)
Classify the following reactions as combination, decomposition, displacement, precipitation and neutralisation. Also balance the equations.
(a) CaCO3 (s) CaO (s) + CO2 (g)
(b) Zn(s) + H2SO4 ⟶ ZnSO4 (s) + H2 (g)
(c) AgNO3 (aq) + NaCl (aq) ⟶ AgCl(s) + NaNO3
(d) NH3 (g) + HCl (g) ⟶ NH4Cl (s)
(e) CuSO4 (aq) + H2S (g) ⟶ CuS (s) + H2SO4 (l)
(f) Zn (s) + CuSO4 (aq) ⟶ ZnSO4 (aq) + Cu(s)
(g) Ca(s) + O2 (g) ⟶ CaO(s)
(h) NaOH + HCl ⟶ NaCl + H2O
(i) KOH + H2SO4 ⟶ K2SO4 + H2O
Answer
(a) Decomposition reaction
CaCO3 (s) CaO (s) + CO2 (g)
(b) Displacement reaction
Zn(s) + H2SO4 ⟶ ZnSO4 (aq) + H2 (g)
(c) Precipitation reaction
AgNO3 (aq) + NaCl (aq) ⟶ AgCl(s) + NaNO3 (aq)
(d) Combination reaction
NH3 (g) + HCl (g) ⟶ NH4Cl (s)
(e) Precipitation reaction
CuSO4 (aq) + H2S (g) ⟶ CuS (s) + H2SO4
(f) Displacement reaction
Zn (s) + CuSO4 (aq) ⟶ ZnSO4 (aq) + Cu(s)
(g) Combination reaction
2Ca(s) + O2 (g) ⟶ 2CaO(s)
(h) Neutralisation reaction
NaOH + HCl ⟶ NaCl + H2O
(i) Neutralisation reaction
2KOH + H2SO4 ⟶ K2SO4 + 2H2O
Write the missing reactants and products and then balance the equations:
(a) NaOH + ............... ⟶ NaCl + ................
(b) KClO3 ................. + ...................
(c) ............. + HCl ⟶ NaCl + H2O + .................
Answer
(a) NaOH + HCl ⟶ NaCl + H2O
(b) 2KClO3 2KCl + 3O2
(c) Na2SO3 + 2HCl ⟶ 2NaCl + H2O + SO2
State the effect of the following on the surroundings.
(a) an endothermic reaction
(b) an exothermic reaction.
Answer
(a) An endothermic reaction causes fall in temperature. These reactions absorb heat from the surroundings leading to cooling effect of the surroundings.
(b) An exothermic reaction causes rise in temperature. These reactions release heat into surroundings leading to increase in temperature of the surroundings.
What do you observe (on the basis of energy) when:
(a) an acid is added to a basic solution?
(b) ammonium chloride is dissolved in water?
Answer
(a) When an acid is added to a basic solution, a neutralisation reaction takes place. Energy is released during the reaction. So, the temperature of the solution rises and the container may feel warm. This is because neutralisation is an exothermic reaction (gives out heat energy).
(b) When ammonium chloride is dissolved in water, heat is absorbed leading to fall in temperature. So the apparatus in which this reaction occurs becomes colder than earlier.
Define :
(a) precipitation
(b) neutralization
(c) catalyst
(d) alkali
Answer
(a) A chemical reaction in which two compounds in their aqueous state react to form an insoluble solid (a precipitate) as one of the products is known as a precipitation reaction.
(b) A chemical reaction in which a base or an alkali reacts with an acid to produce a salt and water only is known as a neutralisation reaction.
(c) A catalyst is a substance that either increases or decreases the rate of a chemical reaction without itself undergoing any chemical change during the reaction.
(d) All metallic oxides and metallic hydroxides are called bases, whereas those bases that dissolve in water are known as alkalis.
What do you observe when
(a) Iron nail is kept in copper sulphate solution for some time?
(b) Phenolphthalein is added to sodium hydroxide solution?
(c) Blue litmus paper is dipped in dilute hydrochloric acid?
(d) Lead nitrate is heated?
(e) Magnesium ribbon is burnt in oxygen?
(f) Ammonia is brought in contact with hydrogen chloride gas?
Answer
(a) When iron nail is kept in a blue coloured copper sulphate solution for sometime, a reddish brown coating is seen on the iron nail and the colour of the solution changes gradually from blue to light green. The reason for this observation is that iron being more reactive than copper, displaces copper from the solution and copper is deposited on the iron nail.
Fe + CuSO4 (aq) ⟶ FeSO4 + Cu
(b) When a few drops of phenolphthalein is added to sodium hydroxide solution, it turns pink indicating that sodium hydroxide solution is basic in nature.
(c) Blue litmus paper changes into red colour when dipped in dilute hydrochloric acid confirming its acidic nature.
(d) When solid lead nitrate is heated strongly, it decomposes to produce light yellow solid lead monoxide, reddish brown nitrogen dioxide gas and colourless oxygen gas.
2Pb(NO3)2 2PbO + 4NO2 (g) + O2 (g)
(e) When magnesium ribbon is burnt in oxygen gas, it burns with a dazzling white light and produces a white powder of magnesium oxide.
2Mg + O2 ⟶ 2MgO
(f) When ammonia is brought in contact with hydrogen chloride gas, dense white fumes of ammonium chloride are produced.
NH3 + HCl ⟶ NH4Cl
Give reason :
A person suffering from acidity is advised to take an antacid.
Answer
Antacids are alkaline in nature. So it neutralises the acid produced in the stomach. Hence, antacids are advised for acidity.
Give reason:
Acidic soil is treated with quicklime.
Answer
Quicklime is a base which neutralises the acidic soil. So acidic soil is treated with base like quicklime.
Give reason:
Wasp sting is treated with vinegar.
Answer
Wasp sting is alkaline in nature which is neutralised by vinegar which is a weak acid.
Explain the following types of chemical reactions giving one example for each of them:
(a) combination reaction
(b) decomposition reaction
(c) displacement reaction
(d) double displacement reaction
Answer
(a) Combination Reaction — A reaction in which two or more substances combine to form a single substance is called a combination reaction. It is also called a synthesis reaction.
Example:
Two elements combine to form a compound.
When iron and sulphur (both elements) are heated together they combine to form a compound iron sulphide.
Fe (s) + S (s) FeS (s)
(b) Decomposition Reaction — A reaction in which a compound breaks up on heating into two or more simpler substances is called a decomposition reaction.
Example:
Calcium carbonate decomposes on strong heating to form two compounds, calcium oxide and carbon dioxide.
CaCO3 (s) CaO (s) + CO2 (g)
(c) Displacement Reaction — A reaction in which a more active element displaces a less active element from a compound is called a displacement reaction.
Example:
Zinc is more reactive than copper so it displaces copper from copper sulphate solution to form zinc sulphate.
Zn (s) + CuSO4 (aq) ⟶ ZnSO4 (aq) + Cu (s)
(d) Double Displacement Reaction — A chemical reaction in which two compounds in their aqueous state exchange their ions or radicals to form new compounds is called a double decomposition reaction.
AB + CD ⟶ CB + AD
Here AB and CD are reactants. They exchange their ions to form CB and AD which are the products.
Example:
When a solution of silver nitrate is added to a solution of sodium chloride a precipitate of silver chloride is formed.
AgNO3 (aq) + NaCl (aq) ⟶ NaNO3 (aq) + AgCl ↓
What are oxides? Give two examples of each of the following oxides.
(a) Basic oxide
(b) Acidic oxide
(c) Amphoteric oxide
(d) Neutral oxide
Answer
Oxides are compounds which essentially contain oxygen in their molecules, chemically combined with a metal or a non-metal.
(a) Calcium oxide (CaO) and magnesium oxide (MgO) are basic oxides.
(b) Carbon dioxide (CO2) and sulphur dioxide (SO2) are acidic oxides.
(c) Zinc oxide (ZnO) and aluminium oxide (Al2O3) are amphoteric oxides.
(d) Carbon monoxide (CO) and nitric oxide (NO) are neutral oxides.
Define exothermic and endothermic reactions. Give two examples of each.
Answer
Exothermic Reaction
A chemical reaction in which heat is given out is called an exothermic reaction.
Examples:
- When carbon burns in oxygen, carbon dioxide is formed and a lot of heat is produced.
C + O2 ⟶ CO2 + Heat - When water is added to quicklime, slaked lime is formed and along with it a large amount of heat energy is produced.
CaO + H2O ⟶ Ca(OH)2 + Heat
Endothermic Reaction
A chemical reaction in which heat is absorbed is called an endothermic reaction.
Examples:
- When nitrogen and oxygen gas are heated together to a temperature of about 3000°C, nitric oxide gas is formed.
N2 + O2 2NO - When calcium carbonate is heated at 1000°C, it decomposes to give calcium oxide and carbon dioxide.
CaCO3 CaO + CO2
How will you obtain?
(a) Magnesium oxide from magnesium.
(b) Silver chloride from silver nitrate.
(c) Nitrogen dioxide from lead nitrate.
(d) Zinc chloride from zinc.
(e) Ammonia from nitrogen.
Also give balanced equations for the reactions.
Answer
(a) When magnesium reacts with oxygen it produces magnesium oxide.
2Mg + O2 ⟶ 2MgO
(b) When silver nitrate reacts with hydrochloric acid silver chloride is formed.
AgNO3 (aq) + HCl ⟶ AgCl↓ + HNO3
(c) When lead nitrate is heated strongly, a reddish brown gas nitrogen dioxide is produced.
2Pb(NO3)2 2PbO + 4NO2 (g) + O2 (g)
(d) Zinc reacts with dilute HCl to produce zinc chloride.
Zn + 2HCl (dil.) ⟶ ZnCl2 + H2 (g)
(e) When nitrogen gas reacts with hydrogen gas in presence of finely divided iron as catalyst, subjected to pressure of 200-900 atm and temperature of about 450°C, ammonia gas is produced.
(a) What is meant by metal activity series?
(b) A metal P was placed in a solution of silver nitrate. After sometime, silver was deposited on metal P and the solution turned blue.
(i) Which is more reactive - metal P or silver?
(ii) What kind of reaction is this?
(iii) Give an example of this type of reaction.
Answer
(a) A list in which the metals are arranged in a decreasing order of their chemical reactivity is called the metal activity series.
(b)(i) Metal P is more reactive as it displaced silver from the solution of silver nitrate, to form new compound.
(ii) In this reaction, metal P displaces silver from its salt (silver nitrate, AgNO3) to form metal P nitrate and silver metal. Hence, the given reaction is a displacement reaction.
(iii) Zinc being more reactive than copper, displaces copper from copper sulphate solution. The blue copper sulphate solution turns colourless due to the formation of zinc sulphate and reddish brown copper is deposited on zinc.
Zn + CuSO4 (aq) → ZnSO4 (aq) + Cu
Following is a list of metals :
Na, Zn, Fe, Ag, Pb, Hg, Cu.
Answer the questions that follow :
(a) Which of the above metals can displace all the metals from their salt solution?
(b) Name one of the above metals which cannot react with dilute acid to produce hydrogen gas.
(c) Name one of the above metals which reacts with oxygen upon heating to produce an amphoteric oxide.
(d) Arrange the above metals in a decreasing order of their reactivity.
Answer
(a) Na, that is Sodium can displace other metals from their salt solutions because it is the most reactive.
(b) Silver (Ag) is very low in the reactivity series, so it does not displace hydrogen from dilute acids.
(c) Zinc (Zn) reacts with oxygen upon heating to produce an amphoteric oxide.
(d) The given metals in a decreasing order of their reactivity are:
Na > Zn > Fe > Pb > Cu > Hg > Ag
Given below is a diagram showing a type of reaction between two compounds. Answer the questions that follow :
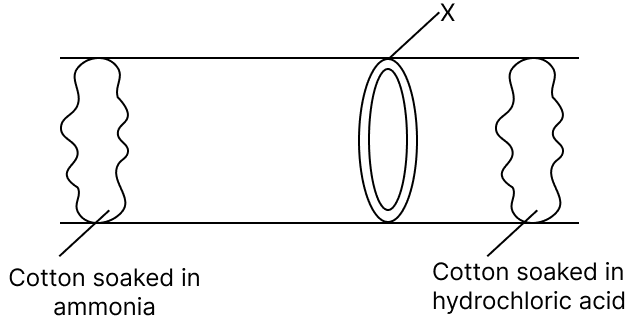
(a) What compound is X?
(b) Give a balanced chemical equation to form X.
(c) Name the type of reaction shown.
Answer
(a) Compound X is Ammonium chloride.
(b) A balanced chemical equation to form Ammonium chloride is:
NH3 (g) + HCl (g) → NH4Cl (s)
(c) The given reaction is a Combination reaction, as two reactants combine to form a new product with new chemical properties.
A teacher showed a chemistry experiment in the class to her students. She poured about 100 ml of sodium hydroxide into a beaker and added a few drops of phenolphthalein to it. The colour of the solution immediately changed. Then she added dilute sulphuric acid to the beaker slowly, while constantly stirring the solution with a glass rod. She kept on adding dilute sulphuric acid until the solution became colourless again. Then she called her students to touch the beaker.
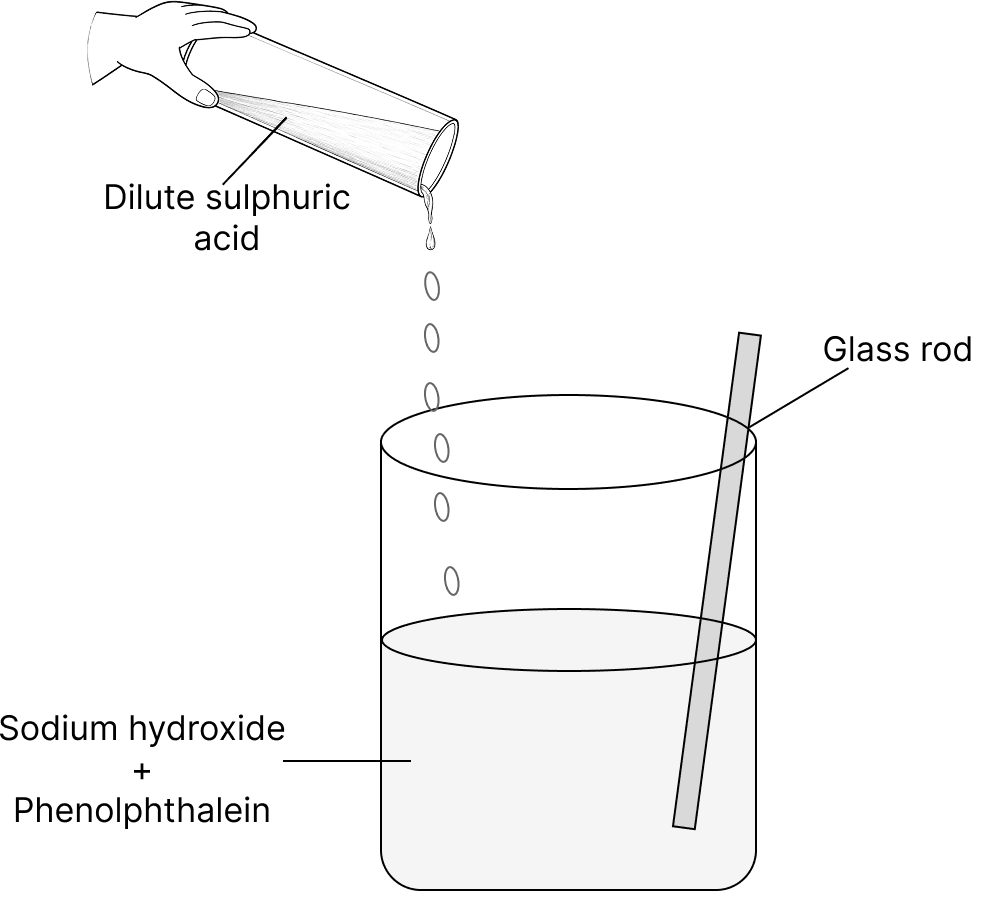
(a) Name the type of reaction which involves an acid and a base as shown by the teacher.
(b) What colour change was observed when phenolphthalein was added to the beaker?
(c) Write a balanced chemical equation for the reaction between sodium hydroxide and dilute sulphuric acid.
(d) What will the students feel on touching the beaker after the reaction? Why?
(e) Why do many toothpastes contain bases?
(f) Name:
(i) a basic substance to treat an acidic soil.
(ii) an acidic substance to treat a basic soil.
Answer
(a) The type of reaction which involves an acid and a base as shown by the teacher is neutralisation reaction.
(b) Phenolphthalein turns pink in a basic solution. When acid is added, the solution becomes colourless.
(c) A balanced chemical equation for the reaction between sodium hydroxide and dilute sulphuric acid is :
2NaOH + H2SO4 ⟶ Na2SO4 + 2H2O
(d) On touching the beaker after the reaction, the students will feel the beaker warm because neutralisation is an exothermic reaction that releases heat.
(e) Many toothpastes contain bases because bases neutralise the acids produced by bacteria in the mouth. This helps to prevent tooth decay.
(f) (i) Quicklime is a basic substance used to treat acidic soil.
(ii) Organic matter or sulphate salts may be used to treat basic soil.