The formula of a compound represents:
- an atom
- a molecule
- an ion
- all of these
Answer
a molecule
Reason — A molecular formula of a compound is the symbolic representation of its (one) molecule. It shows the number of atoms of each element present in it. These atoms combine in whole numbers to form the molecule.
The symbol of sulphite ion is:
- SO42-
- HSO4-
- SO32-
- S2-
Answer
SO32-
Reason — The symbol of sulphite ion is SO32-.
Which of the following sets of elements show variable valencies?
- Iron and calcium
- Copper and sodium
- Lead and mercury
- Silver and potassium
Answer
Lead and mercury
Reason — Lead has a variable valency of 2 and 4, while mercury has a variable valency of 1 and 2.
Which of the following has zero valency?
- Hydrogen
- Chlorine
- Carbon
- Neon
Answer
Neon
Reason — Neon is the element with zero valency, as it doesn't form bonds under normal conditions.
The valencies shown by copper is:
- 1 and 2
- 2 and 3
- 2 and 4
- 1 and 3
Answer
1 and 2
Reason — The valencies shown by copper are 1 and 2.
Which of the following represents the correct symbol of potassium ion ?
- K
- K+
- K2+
- K3+
Answer
K+
Reason — The correct symbol for the potassium ion is K+.
Assertion (A): When an atom gains one or more electrons, it becomes negatively charged.
Reason (R): The number of electrons in an atom exceeds the number of protons upon gaining electrons.
- Both A and R are true and R is the correct explanation of A.
- Both A and R are true but R is not the correct explanation of A.
- A is true but R is false.
- A is false but R is true.
Answer
Both A and R are true and R is the correct explanation of A.
Explanation — In a neutral atom, the number of electrons and protons are equal, so they are electrically neutral. However, when an atom gains one or more electrons, the number of electrons in an atom exceeds the number of protons, and the atom becomes negatively charged.
Assertion (A): Each element has its own unique symbol, which is a short hand representation of its atom.
Reason (R): The symbol of some elements is the same as the first letter of their names.
- Both A and R are true and R is the correct explanation of A.
- Both A and R are true but R is not the correct explanation of A.
- A is true but R is false.
- A is false but R is true.
Answer
Both A and R are true but R is not the correct explanation of A.
Explanation — The symbol can be based on the Latin or Greek name of the element, or a combination of letters, but it’s not strictly the first letter. For example, Helium has the symbol He, Sodium has Na, and Iron has Fe, which are not simply the first letter of the element's name.
Assertion (A): Magnesium burns in oxygen to produce a compound magnesium oxide with the molecular formula MgO.
Reason (R): Magnesium oxide is a white amorphous solid.
- Both A and R are true and R is the correct explanation of A.
- Both A and R are true but R is not the correct explanation of A.
- A is true but R is false.
- A is false but R is true.
Answer
Both A and R are true but R is not the correct explanation of A.
Explanation — Magnesium burns in oxygen to form magnesium oxide, whose molecular formula is MgO. Magnesium oxide is a white amorphous solid. However, the fact that magnesium oxide is a white amorphous solid does not explain why magnesium burns in oxygen to form MgO.
Assertion (A): The valency of inert gases, helium, neon and argon is zero.
Reason (R): The outermost shell of inert gases is complete.
- Both A and R are true and R is the correct explanation of A.
- Both A and R are true but R is not the correct explanation of A.
- A is true but R is false.
- A is false but R is true.
Answer
Both A and R are true and R is the correct explanation of A.
Explanation — The valency of inert gases, helium, neon and argon is zero as the outermost shell of inert gases is complete.
Assertion (A): A chemical equation is the symbolic representation of a chemical reaction using symbols and formulae of the substances involved in the reaction.
Reason (R): Every chemical equation needs to be balanced to follow the law of conservation of mass.
- Both A and R are true and R is the correct explanation of A.
- Both A and R are true but R is not the correct explanation of A.
- A is true but R is false.
- A is false but R is true.
Answer
Both A and R are true but R is not the correct explanation of A.
Explanation — A chemical equation is the symbolic representation of a chemical reaction using symbols and formulae of the substances involved in the reaction, and every chemical equation needs to be balanced to follow the law of conservation of mass. However, they are two separate facts.
Fill in the blanks:
(a) A symbol represents ............... of an element.
(b) An atom is neither created nor destroyed during a ............... .
(c) ............... are positively charged ions.
(d) Total number of atoms of all the elements present in Al2O3 is ............... .
(e) A ............... is the symbolic representation of a chemical reaction using symbols and formulae of the substances taking part in the reaction.
Answer
(a) atom
(b) chemical reaction
(c) Cations
(d) five
(e) chemical equation
Write 'true' or 'false' for the following and correct the false statements.
(a) When an atom gains one or more electrons, it becomes positively charged.
(b) The valency of nitrogen in ammonia gas is 3.
(c) The sulphide ion is a polyatomic ion.
(d) The nitrate ion is represented as NO2-.
(e) The element cobalt is represented as CO.
Answer
(a) False
Correct Statement — When an atom gains one or more electrons, it becomes negatively charged.
(b) True
(c) False
Correct Statement — The sulphide ion (S2-) is a monoatomic ion.
(d) False
Correct Statement — The nitrate ion is represented as NO3-.
(e) False
Correct Statement — The element cobalt is represented as Co.
Match the following:
| Column A | Column B |
|---|---|
| (a) Combining capacity of an element | (i) Silicon dioxide |
| (b) The radical/ion formed due to the loss of electrons | (ii) Molecular formula |
| (c) Symbolic representation of a compound | (iii) Calcium carbonate |
| (d) Marble | (iv) Valency |
| (e) Sand | (v) Cation |
Answer
| Column A | Column B |
|---|---|
| (a) Combining capacity of an element | (iv) Valency |
| (b) The radical/ion formed due to the loss of electrons | (v) Cation |
| (c) Symbolic representation of a compound | (ii) Molecular formula |
| (d) Marble | (iii) Calcium carbonate |
| (e) Sand | (i) Silicon dioxide |
Name the following:
(a) A polyatomic positive ion.
(b) The substances written on the left side of an arrow in an equation.
(c) The substances which are formed during a chemical reaction.
(d) An unbalanced chemical equation.
(e) An atom or group of atoms that behave as a single charged unit.
(f) A substance used to increase the rate of reaction.
Answer
(a) Ammonium ion, NH4+
(b) Reactants
(c) Products
(d) Skeletal equation
(e) Radical
(f) Catalyst
Write the names of the following ions:
(a) HCO3-
(b) MnO4-
(c) N3-
(d) Cr2O72-
Answer
(a) Bicarbonate
(b) Permanganate
(c) Nitride
(d) Dichromate
Write the names of the compounds represented by the following formulae :
(a) K2CO3
(b) P2O5
(c) H2S
(d) Ca(OH)2
(e) CuSO4
(f) SiO2
Answer
(a) Potassium carbonate
(b) Phosphorus pentoxide
(c) Hydrogen sulphide
(d) Calcium hydroxide
(e) Copper sulphate
(f) Silicon dioxide
Give the symbols and valencies of the following radicals:
(a) Hydroxide
(b) Chloride
(c) Carbonate
(d) Ammonium
(e) Nitrate
Answer
| Radical | Symbol | Valency |
|---|---|---|
| Hydroxide | OH- | 1 |
| Chloride | Cl- | 1 |
| Carbonate | CO32- | 2 |
| Ammonium | NH4+ | 1 |
| Nitrate | NO3- | 1 |
Write the molecular formulae for the following compounds and name the elements present.
(a) Sodium bicarbonate
(b) Sodium chloride
(c) Sulphuric acid
(d) Nitric acid
Answer
| Compounds | Molecular formula | Elements present |
|---|---|---|
| Sodium bicarbonate | NaHCO3 | Sodium, Hydrogen, Carbon, Oxygen |
| Sodium chloride | NaCl | Sodium, Chlorine |
| Sulphuric acid | H2SO4 | Hydrogen, Sulphur, Oxygen |
| Nitric acid | HNO3 | Hydrogen, Nitrogen, Oxygen |
The valency of aluminium is 3. Write the valency of other radicals present in the following compounds. Also name the compounds
(a) AlCl3
(b) Al2O3
(c) AlN
(d) Al2(SO4)3
Answer
| Compounds | Radical | Valency |
|---|---|---|
| Aluminium chloride (AlCl3) | Chloride (Cl-) | 1 |
| Aluminium oxide (Al2O3) | Oxide (O2-) | 2 |
| Aluminium nitride (AlN) | Nitride (N3-) | 3 |
| Aluminium sulphate (Al2(SO4)3) | Sulphate (SO42-) | 2 |
Write the molecular formulae for the oxides and sulphides of the following elements.
(a) Sodium
(b) Calcium
(c) Hydrogen
Answer
| Element | Oxide | Sulphide |
|---|---|---|
| Sodium | Na2O | Na2S |
| Calcium | CaO | CaS |
| Hydrogen | H2O | H2S |
Define:
(a) Symbol
(b) Molecular formula
(c) Radical
(d) Valency
Answer
(a) A symbol is the short hand representation for the atom of a specific element. Each chemical element has its own unique symbol. For example, H represents Hydrogen, and O represents Oxygen.
(b) A molecular formula of a compound is the symbolic representation of its one molecule which shows the number of atoms of each element present in it.
(c) Radical is an atom of an element or a group of atoms of the same or different elements that behave as a single unit and has positive or negative charge.
(d) Valency is the combining capacity of an atom of an element or of a radical with the atoms of other elements or radicals to form molecules.
Differentiate between:
(a) Reactants and Products
(b) Cation and anion
(c) A balanced and an unbalanced chemical equation
Answer
(a) Differences between Reactants and Products
| Reactants | Products |
|---|---|
| The substances that are used as the starting material and which react with one another are called reactants. | The substances which are formed as a result of the reaction are called products. |
| Reactants are written on the left hand side of the equation. | Products are written on the right hand side of the equation. |
| In equation C + O2 ⟶ CO2, C and O2 are reactants. | In equation C + O2 ⟶ CO2, CO2 is product. |
(b) Differences between Cation and anion
| Cation | Anion |
|---|---|
| A cation is a positively charged ion. | An anion is a negatively charged ion. |
| A cation is formed when an atom loses electrons. | An anion is formed when an atom gains electrons. |
| Example: Na+ (sodium ion), Ca2+ (calcium ion). | Example: Cl- (chloride ion), OH- (hydroxide ion). |
(c) Differences between a balanced and an unbalanced chemical equation
| Balanced chemical equation | Unbalanced chemical equation |
|---|---|
| In a balanced chemical equation the total number of atoms on the reactant side as well as the product side are equal. | In an unbalanced chemical equation the total number of atoms on the reactant side and the product side are not equal. |
| Example: Zn + H2SO4 ⟶ ZnSO4 + H2 | Example: H2 + O2 ⟶ H2O |
State the law of conservation of mass.
Answer
The law of conservation of mass states that matter can neither be created nor be destroyed in a chemical reaction. Only it can be changed from one form to another during the reaction process.
State two points of information necessary to write the molecular formula of a compound.
Answer
To write the molecular formula of a compound, the following information should be available:
- Symbols of the elements or the radicals that constitute the compound.
- Valencies (combining capacities) of the elements or the radicals.
Balance the following equations:
N2 + H2 ⟶ NH3
H2 + O2 ⟶ H2O
Na2O + H2O ⟶ NaOH
CO + O2 ⟶ CO2
Zn + HCl ⟶ ZnCl2 + H2
Answer
N2 + 3H2 ⟶ 2NH3
2H2 + O2 ⟶ 2H2O
Na2O + H2O ⟶ 2NaOH
2CO + O2 ⟶ 2CO2
Zn + 2HCl ⟶ ZnCl2 + H2
Write balanced chemical equations using symbols and formulae for the following word equations:
Iron + Chlorine ⟶ Iron(III) chloride
Magnesium + dil. sulphuric acid ⟶ Magnesium sulphate + hydrogen
Magnesium + oxygen ⟶ Magnesium oxide
Calcium oxide + water ⟶ Calcium hydroxide
Sodium + Chlorine ⟶ Sodium chloride
Answer
2Fe + 3Cl2 ⟶ 2FeCl3
Mg + H2SO4 ⟶ MgSO4 + H2
2Mg + O2 ⟶ 2MgO
CaO + H2O ⟶ Ca(OH)2
2Na + Cl2 ⟶ 2NaCl
What information do you get from the following chemical equations?
(a) Zn(s) + 2HCl (dil) ⟶ ZnCl2 (aq) + H2 (g)
(b) N2(g) + 3H2(g) 2NH3(g) + Heat
Answer
(a) We get the following information from the given chemical equation:
- Zinc and Hydrochloric acid are reactants. Zinc is in solid state that reacts with dilute HCl.
- The reaction is irreversible.
- The products formed are an aqueous solution of zinc chloride and hydrogen gas.
- The equation is balanced i.e., number of atoms of each element on the reactant side and the product side are equal.
(b) We get the following information from the given chemical equation:
- The reactants, nitrogen and hydrogen, and the product, ammonia, are all gases.
- The reaction is reversible and exothermic.
- The reaction takes place at a temperature between 400-450°C and at a high pressure between 200-900 atmosphere.
- The catalyst is iron and molybdenum acts as a promoter, which increases the efficiency of the catalyst.
- The equation is balanced i.e., number of atoms of each element on the reactant side and the product side are equal.
What is variable valency? Give two examples of elements showing variable valency.
Answer
Some elements exhibit more than one valency and they are said to have variable valency.
Examples: Iron and copper.
| Metal | Radicals | Valency |
|---|---|---|
| Iron (Fe) | Ferrous, Ferric | 2, 3 |
| Copper (Cu) | Cuprous, Cupric | 1, 2 |
(a) What is a chemical equation?
(b) Why is it necessary to balance a chemical equation?
(c) What are the limitations of a chemical equation?
Answer
(a) A chemical equation is the symbolic representation of a chemical reaction using symbols and formulae of the substances involved in the reaction.
(b) It is necessary to balance a chemical equation so as to make the number of atoms of the reactants equal to the number of atoms of the products because a chemical reaction is just a rearrangement of atoms and atoms are neither created nor destroyed during the chemical reaction.
(c) The limitations of a chemical equation are:
It does not inform about:
- the physical states of the reactants and the products i.e. whether they are solids, liquids or gases.
- the concentration of reactants and products.
- the time taken for the completion of reaction.
- the rate at which a reaction proceeds.
- the heat changes during the reaction i.e. whether heat is given out or absorbed.
- the conditions such as temperature, pressure, catalyst, etc. which affect the reaction.
- the nature of the reaction i.e. whether it is reversible or irreversible.
State the important information obtained from the following in four points:
(a) A molecular formula of a compound.
(b) A balanced chemical equation for a reaction.
Answer
(a) The molecular formula of a compound gives the following information:
- It represents one molecule of a compound.
- The elements or ions present in the compound.
- The number of each kind of atoms and ions present in one molecule of a compound and their ratio.
- The molecular formula enables us to calculate the mass of one molecule of the compound if the atomic mass of each atom present in the molecule is known. Molecular mass is the algebraic sum of the masses of all the atoms present in a given molecule.
(b) A balanced chemical equation gives the following information:
- It shows which substances are taking part in a chemical reaction and what products are obtained as a result of it (Qualitative).
- It shows both the number of molecules and the number of atoms of each type involved in the reaction.
- It enables us to calculate the actual amount (mass) of reactants and products involved and formed in the reaction if atomic mass of each of the elements involved in the reaction is known (Quantitative).
- It makes the study of chemistry universally standardized.
State five ways by which a chemical equation can be made more informative.
Answer
A chemical equation can be made more informative in the following ways:
- The physical state of the reactants and products can be indicated by putting (s) for solid, (l) for liquid, (g) for gas and (aq.) for aqueous state beside the symbols for the reactants and products.
- Evolution or absorption of heat during the reaction can be denoted by adding or subtracting a heat term on the product side.
- Temperature, pressure and catalyst can be indicated above or below the arrow (→) separating the reactants and products.
- Concentrations of reactants and products are indicated by adding the words (dil.) for dilute and (conc.) for concentrated before their formulae.
- By using the signs → and ⇌, information about irreversible and reversible reactions can be depicted.
Given below are two groups showing basic and acid radicals. Based on these two groups, answer the questions that follow :
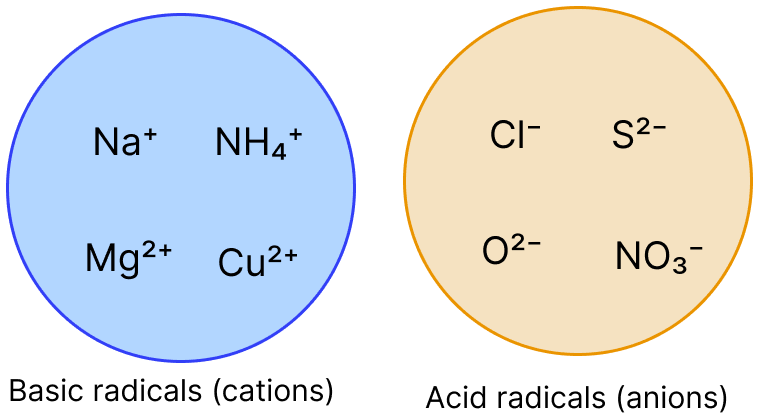
(a) Which basic radical or cation exhibits variable valency ?
(b) How is NH4+ ion different from other basic radicals in the group ?
(c) Which acid radical or anion is the odd one out in the group ? Justify your answer.
(d) Using one radical from each group, form a compound that is commonly used in daily life. Write its name and chemical formula.
Answer
(a) Copper exhibits variable valency. It shows valencies 1 and 2.
(b) NH4+ ion is different from the other basic radicals because it is a polyatomic ion, whereas the others are simple metal ions.
(c) NO3- is the odd one out because it is the only polyatomic acid radical in the group. Cl-, S2- and O2- are monoatomic ions.
(d) Using Na+ from basic radicals and Cl- from acid radicals, the compound formed is sodium chloride. Its chemical formula is NaCl.