Chemistry
A student was asked to perform two experiments in the laboratory based on the instructions given:
Observe the picture given below and state one observation for each of the Experiments 1 and 2 that you would notice on mixing the given solutions.
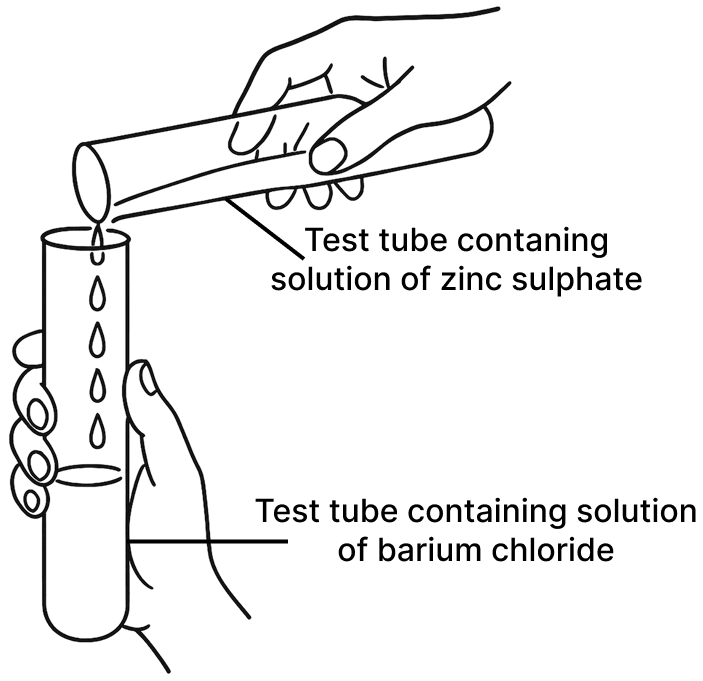
(a) Experiment 1
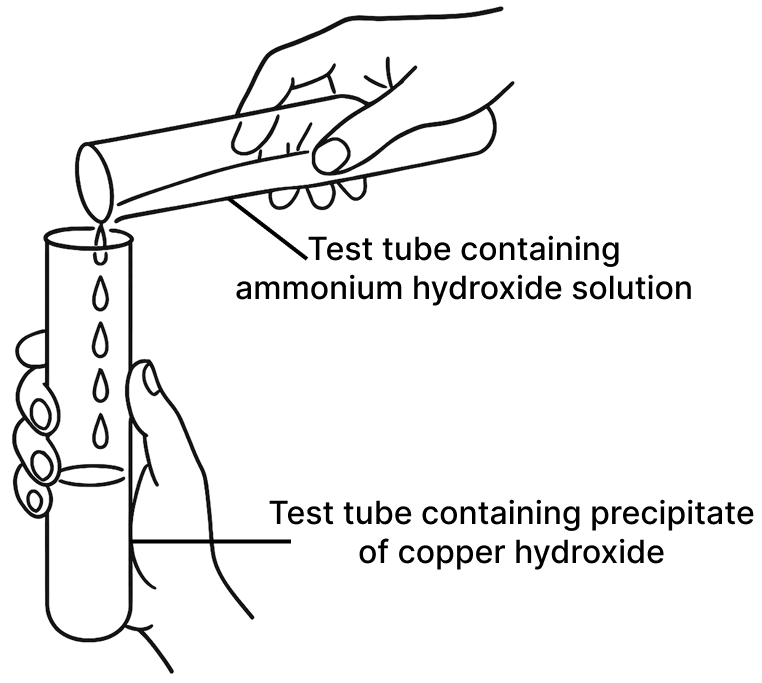
(b) Experiment 2
Answer
Experiment 1:
Mixing Zinc Sulphate and Barium Chloride
Observation: A white precipitate is formed immediately upon mixing the two colourless solutions.
Explanation: This is a double displacement reaction. The Barium ions Ba2+ react with the Sulphate ions SO42- to form Barium Sulphate BaSO4, which is an insoluble white solid.
Experiment 2:
Adding Ammonium Hydroxide to Copper Hydroxide
Observation: The pale blue precipitate of copper hydroxide dissolves to form a deep blue (inky blue) solution.
Explanation: Copper(II) hydroxide reacts with excess ammonium hydroxide to form a soluble complex called Tetraammine copper(II) hydroxide. This complex is responsible for the characteristic deep blue color.
\underset{\text{pale blue precipitate}}{\text{Cu(OH)}2{(aq)}} + \underset{\text{colourless solution}}{4\text{NH}4\text{OH{(aq)} (excess)}} \longrightarrow \underset{\text{deep blue solution}}{[\text{Cu(NH}3)4](\text{OH})2{(aq)}} + \underset{\text{water}}{4\text{H}2\text{O}}
Related Questions
Given below in column A is a schematic diagram of the electrolytic reduction of alumina. Identify the parts labelled as A, B and C with the correct options from the Column B.

Element 'X' forms an oxide with the formula X2O3 which is a solid with high melting point. 'X' would most likely be placed in the group of the Periodic Table as:
(a) Na
(b) Mg
(c) Al
(d) SiJustify your answer in the above question (1).
Copper sulphate solution is electrolysed using copper electrodes.
(a) Which electrode [cathode or anode] is the oxidizing electrode? Why?
(b) Write the equation for the reaction occurring at the above electrode.
X [2, 8, 7] and Y [2, 8, 2] are two elements. Using this information complete the following:
(a) …………… is the metallic element.
(b) Metal atoms tend to have a maximum of …………… electrons in the outermost shell.
(c) …………… is the reducing agent.