Chemistry
Complete the following by choosing the correct answers from the bracket:
(a) When dilute sulphuric acid reacts with zinc granules, the gas evolved is …………… (hydrogen / carbon dioxide), which can be tested using a burning splint.
(b) A solution of copper(II) sulphate in sodium hydroxide solution forms a …………… (pale blue / green) precipitate.
(c) In methane, each hydrogen atom share(s) …………… (one / two) electron(s) with the central carbon atom to complete its valence shell.
(d) The electron affinity of element X is greater than that of element Y. The oxidising power of X is likely to be …………… (more / less) than that of element Y.
(e) The naturally occurring compound of a metal from which the metal can be extracted is called its …………… (ore / mineral).
Sulphuric Acid
18 Likes
Answer
(a) When dilute sulphuric acid reacts with zinc granules, the gas evolved is hydrogen, which can be tested using a burning splint.
(b) A solution of copper(II) sulphate in sodium hydroxide solution forms a pale blue precipitate.
(c) In methane, each hydrogen atom share one electron with the central carbon atom to complete its valence shell.
(d) The electron affinity of element X is greater than that of element Y.The oxidising power of X is likely to be more than that of element Y.
(e) The naturally occurring compound of a metal from which the metal can be extracted is called its ore.
Answered By
10 Likes
Related Questions
Electroplating steel objects with silver involves a three-step process.
Step 1 — A coating of copper is applied to the object.
Step 2 — A coating of nickel is applied to the object.
Step 3 — The coating of silver is applied to the object.
(a) A diagram of the apparatus used for step 1 is shown.

The chemical process taking place on the surface of the object is
Cu2+(aq) + 2e- ⟶ Cu (s)
What is the observation seen on the surface of the object?Explain why the concentration of copper ions in the electrolyte remains constant throughout step 1.
(b) Give two changes which would be needed in order to coat nickel onto the object in step 2.
(c) Write down the reaction taking place at the positive electrode during step 3.
Identify the following:
(a) A bond formed between two atoms by sharing of a pair of electrons, with both electrons being provided by the same atom.
(b) A salt formed by the complete neutralization of an acid by a base.
(c) A reaction in which the hydrogen of an alkane is replaced by a halogen.
(d) The energy required to remove an electron from a neutral gaseous atom.
(e) A homogenous mixture of two or more metals or a metal and a non-metal in a definite proportion in their molten state.
Match the following Column A with Column B.
Column A Column B (a) Aluminium 1. Covalent compound (b) Sulphuric acid 2. Carbonate ore (c) Calcination 3. Hall Heroult's process (d) Calcium Chloride 4. Contact Process (e) Carbon tetrachloride 5. Electrovalent compound (a) Give the IUPAC name of the following organic compounds:
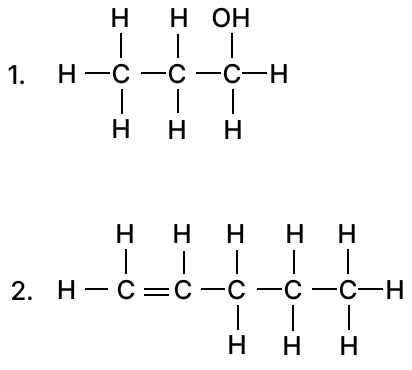
(b) Draw the structural diagram for the following compounds:
- but-2-yne
- 1, 1, 1, trichloro methane
- pentan-2-ol