Chemistry
Explain why thick white fumes are formed when a glass rod dipped in NH4OH is brought near the mouth of a bottle full of HCl gas.
Hydrogen Chloride
85 Likes
Answer
When a glass rod dipped in ammonium hydroxide (NH4OH) is brought near the mouth of a bottle full of HCl gas, then it leads to the formation of dense white fumes due to the production of ammonium chloride.
NH4OH + HCl ⟶ NH4Cl + H2O
Answered By
60 Likes
Related Questions
Study the figure given below and answer the questions that follow :

(i) Identify the gas Y.
(ii) What property of gas Y does this experiment demonstrate?
(iii) Name another gas which has the same property and can be demonstrated through this experiment.
Identify the gas evolved and give the chemical test in each of the following cases.
Dilute hydrochloric acid reacts with:
(i) Iron [II] sulphide.
(ii) Sodium sulphite.
State your observations:
(a) HCl gas is passed through lead nitrate solution and the mixture is heated.
(b) Hydrochloric acid is added to silver nitrate solution.
(c) Ammonium hydroxide solution is added to the resultant product of part (b).
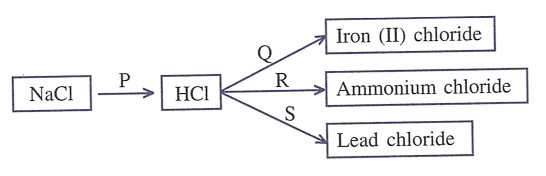
Refer to the diagram given below and write balanced equations with conditions, if any, for the following conversions P to S.