Chemistry
How are the metals like sodium, potassium and calcium obtained. Give equations.
Metallurgy
47 Likes
Answer
Oxides of highly active metals like sodium, potassium and calcium are obtained by electrolytic reduction of fused metallic salts (halides and oxides) using inert electrodes.
Sodium
Electrolyte : Fused sodium chloride
Reaction : NaCl ⇌ Na+ + Cl-
Reaction at cathode : Na+ + e- ⟶ Na
Reaction at anode : Cl- - e- ⟶ Cl
Cl + Cl ⟶ Cl2
Potassium
Electrolyte : Fused potassium bromide
Reaction : KBr ⇌ K+ + Br-
Reaction at cathode : K+ + e- ⟶ K
Reaction at anode : Br- - e- ⟶ Br
Br + Br ⟶ Br2
Calcium
Electrolyte : Fused calcium chloride
Reaction : CaCl2 ⇌ Ca2+ + 2Cl-
Reaction at cathode : Ca2+ + 2e- ⟶ Ca
Reaction at anode : Cl- - e- ⟶ Cl
Cl + Cl ⟶ Cl2
Answered By
31 Likes
Related Questions
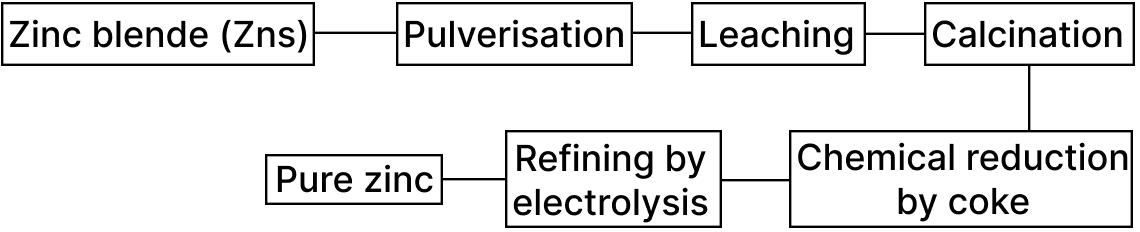
The following flow chart shows the process of extraction of zinc. Out of the 5 steps, 2 steps are incorrect. Identify and correct them.
State why aluminium is extracted from it's oxide by electrolysis while copper, lead, iron by reducing agents and mercury and silver by thermal decomposition.
(a) On which factors does purification of metals depend?
(b) Name the methods used for purification.
(c) With a labelled diagram explain electro-refining of a particular metal.
State the position of aluminium in the periodic table.