Chemistry
Select the correct statement(s) for the diagram shown below.
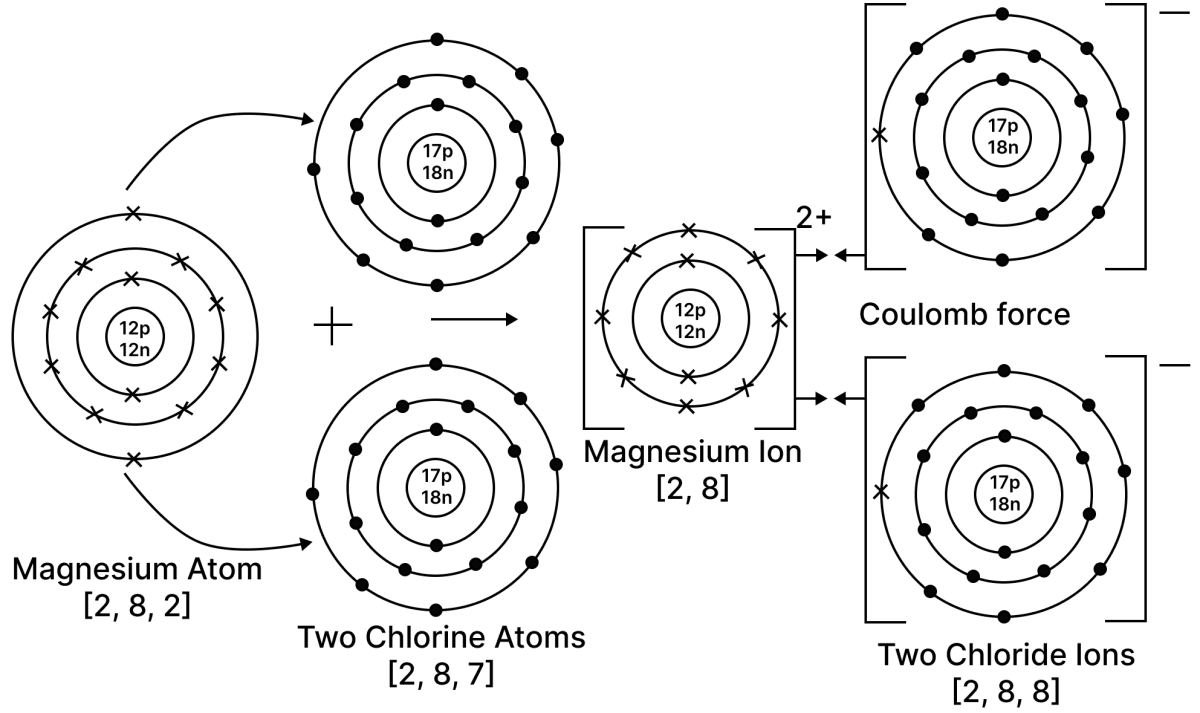
P — Mg loses electrons and undergoes reduction.
Q — Cl gains electrons and undergoes reduction.
R — Mg loses electrons and undergoes oxidation.
- Only P
- Only Q
- Only R
- Both Q and R
Answer
Both Q and R
Reason — Magnesium has two valence electrons in its outermost orbit. So, it will donate its electrons and undergo oxidation to obtain octet configuration. Each chlorine has 7 valence electrons in its outermost orbit. So, it will gain one electron each and undergo reduction to obtain octet configuration.
Related Questions
Postulates of modern atomic theory are:
P — Atoms of an elements may not be alike.
Q — Atoms of elements combine in small whole numbers to form molecules.
R — Atoms can neither be created nor destroyed.
- Only P
- Only Q
- Only R
- Both P and Q
An atom is electrically neutral because :
P — Electrons, protons and neutrons are equal in number.
Q — Protons are equal to neutrons.
R — Protons are equal to electrons.
- Only P
- Only Q
- Only R
- Both Q and R
Select the correct statement(s) for the diagram shown below.
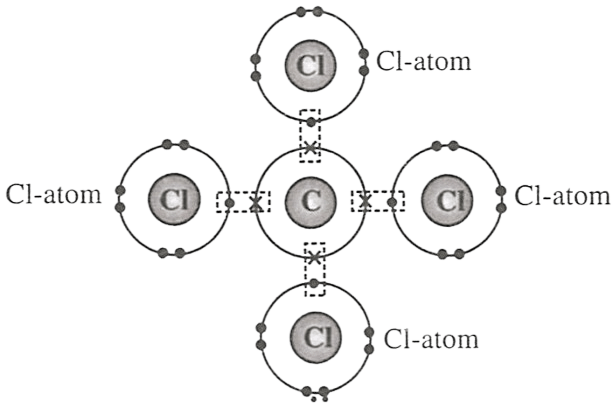
P — One atom of carbon transfers one electron to each chlorine atom.
Q — One atom of carbon shares four electron pairs, one with each of the four atoms of chlorine.
R — Carbon atom attains neon configuration and chlorine attains argon configuration after the combination.
- Only P
- Only Q
- Only R
- Both Q and R
Match the atomic numbers 4, 8, 10, 15 and 19 with each of the following:
(a) Element which can form trivalent ion.
(b) An element with 4 shells.
(c) Element with 6 valence electrons.
(d) Element which does not form an ion.