The energy which flows from a hot body to a cold body is called :
- mechanical energy
- fuel energy
- solar energy
- heat energy
Answer
heat energy
Reason — Heat is the form of energy that is transferred between two bodies at two different temperatures when kept in contact.
1 Joule is equal to :
- 106 erg
- 105 erg
- 107 erg
- 109 erg
Answer
107 erg
Reason — The S.I. unit of heat is joule and its C.G.S. unit is erg, where 1 Joule = 107 erg
............... determines the direction of flow of heat.
- Heat
- Temperature
- Coldness
- None of these
Answer
Temperature
Reason — Temperature is a quantity which indicates the thermal state of a body (i.e., the degree of hotness or coolness of the body). It determines the direction of flow of heat when two bodies at different temperatures are placed in contact.
The S.I. unit of temperature is Kelvin (K).
The amount of heat energy contained by a body depends on :
- the mass of the body
- the temperature of the body
- the nature of the material of the body
- all of the above
Answer
all of the above
Reason — The amount of heat energy contained by a body depends on its mass, temperature and the nature of the material of the bdy.
Celsius and Kelvin scales are related as :
Answer
Reason —
Thus, by adding 273 to the temperature in degree celsius, we get the temperature in Kelvin.
The temperature of a body depends on the average ............... of the molecules.
- potential energy
- kinetic energy
- elastic energy
- atomic energy
Answer
kinetic energy
Reason — The temperature rises when the kinetic energy of the molecules of the substance increases and vice versa. Hence, the temperature of a body depends on the average kinetic energy of the molecules.
The temperature at which pressure and volume of a gas becomes zero is :
- 0 °C
- 0 K
- 0 °F
- 273 K
Answer
0 K
Reason — On Kelvin scale, 0 K is the temperature at which pressure and volume of a gas becomes zero. 0 K is called absolute zero. Thus, negative temperature is not possible on kelvin scale.
The steam point on Fahrenheit scale is :
- 273 °F
- 100 °F
- 213 °F
- 212 °F
Answer
212 °F
Reason — The steam point is 100 °C on Celsius scale, 212 °F on Fahrenheit scale and 373 K on Kelvin scale.
Celsius and Fahrenheit scales are related as :
=
=
5C =
=
Answer
=
Reason — The temperature on Celsius and Fahrenheit scales are realted as :
The increase in the length of a solid on heating is called :
- contraction
- linear expansion
- cubical expansion
- superficial expansion
Answer
linear expansion
Reason — A solid has a definite shape, so when it is heated, it expands in all directions i.e., the length, area and volume all increase on heating. The increase in the length is called linear expansion.
On heating, liquids expand ............... than solids and gases expand ............... than liquids.
- more, less
- less, more
- more, much more
- less, less
Answer
more, much more
Reason — Liquids and gases do not have have a definite shape, so they have only the cubical (or volumetric expansion). On heating, liquids expand more than solids and gases expand much more than liquids.
Water is cooled from 4 °C to 0 °C. It :
- contracts
- expands
- first contracts, then expands
- first expands, then contracts.
Answer
expands
Reason — The expansion of water when it is cooled from 4 °C to 0 °C, is known as anomalous expansion of water.
The change in volume of water when it is cooled from ...............°C to ...............°C is known as anomalous expansion of water.
- 0, 4
- 4, 0
- 1, 0
- 0, 1
Answer
4, 0
Reason — The expansion of water when it is cooled from 4 °C to 0 °C, is known as anomalous expansion of water.
The property of ice which prevents the flow of heat from water of a pond (or lake) to the atmosphere is :
- conduction
- insulation
- low specific heat
- none of the above
Answer
insulation
Reason — Ice is a poor conductor of heat. Hence, it acts as an insulator and prevents the flow of heat from water of a pond (or lake) to the atmosphere.
Identify the correct graph showing the variation in the density of water with temperature :
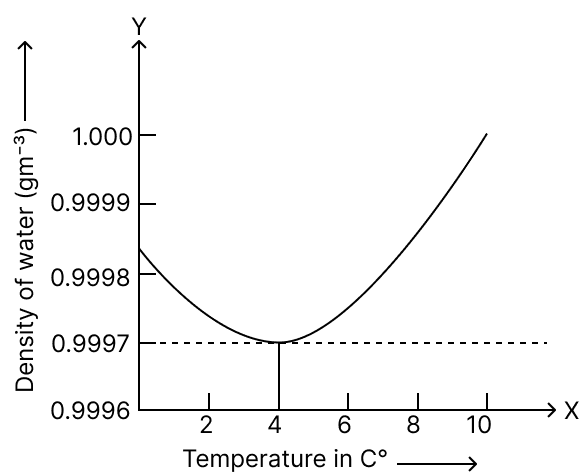
Answer

Reason — The density of water first increases on heating it from 0 °C to 4 °C and then decreases on further heating it from 4 °C to 10 °C. The density of water is thus maximum at 4 °C.
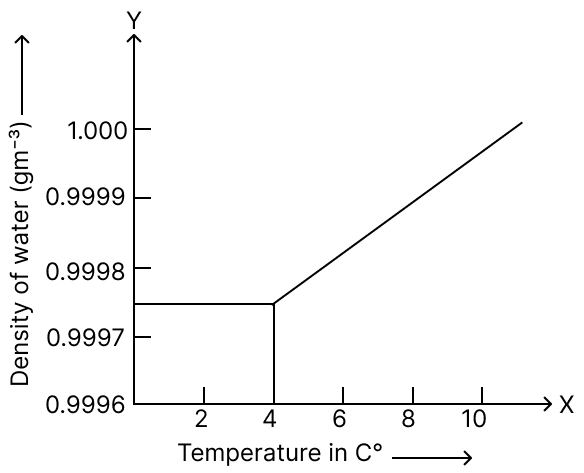
Some quantity of water at 0 °C is heated to 20 °С. Identify the correct graph showing the variation of volume of water with temperature.
-exc-6a-icse-9-phy-concise-485x377.png)
-exc-6a-icse-9-phy-concise-447x379.png)
-exc-6a-icse-9-phy-concise-431x356.png)
-exc-6a-icse-9-phy-concise-506x405.png)
Answer
-exc-6a-icse-9-phy-concise-506x405.png)
option 4
Reason — As from 0 °C to 4 °C, water contracts so its volume decreases as temperature increases, at 4 °C water has maximum density and minimum volume and from 4 °C to 20 °C, water expands, i.e., volume increases as temperature increases.
So, the correct graph should show a decrease in volume from 0 °C to 4 °C and then an increase in volume from 4 °C to 20 °C. This forms a "U" shaped curve with the minimum point at 4 °C.
What is heat? Write its S.I. unit.
Answer
Heat is the form of energy that is transferred between two bodies at two different temperatures when kept in contact.
The S.I. unit of heat is joule (J).
Two bodies at different temperatures are placed in contact. State the direction in which heat will flow.
Answer
The flow of heat is from the body at a higher temperature to a body at a lower temperature. So, when a hot body is kept in contact with a cold body, the hot body becomes less hot and the cold body becomes less cold.
Name the S.I. unit of heat. How is it related to the unit calorie?
Answer
The S.I. unit of heat is joule (J).
1 J = 0.24 cal (approximately).
Define temperature and write its S.I. unit.
Answer
Temperature is a quantity which indicates the thermal state of a body (i.e., the degree of hotness or coolness of the body). It determines the direction of flow of heat when two bodies at different temperatures are placed in contact.
The S.I. unit of temperature is Kelvin (K)
Why does a piece of ice when touched with hand, appear cool? Explain.
Answer
When we touch a piece of ice with our hand, heat passes from our hand to the ice. This happens because flow of heat is from a hot body to a cold body and as our hand is at a higher temperature than the ice cube therefore heat passes from our hand to the ice and we feel cold.
Name two substances which expand on heating.
Answer
The two substances that expand on heating are Brass and Iron.
Name two substances which contract on heating.
Answer
The two substances that contract on heating are —
- Water from 0 °C to 4 °C
- Silver iodide from 80 °C to 141 °C
What do you mean by anomalous expansion of water?
Answer
The expansion of water when it is cooled from 4 °C to 0 °C, is known as anomalous expansion of water.
At what temperature the density of water is maximum? State its value.
Answer
The density of water is maximum at 4 °C. Its value is 1000 kg m-3.
Deep pond of water has its top layer frozen during water. State the expected temperature of water layer (i) just in contact with ice, (ii) at the bottom of pond.
Answer
(i) The temperature of water layer just in contact with ice is 0 °C.
(ii) The temperature of water layer at the bottom of pond is 4 °C.
Distinguish between heat and temperature.
Answer
| Heat | Temperature |
|---|---|
| Heat is a form of energy obtained due to random motion of molecules in a system. | Temperature is a quality which tells the thermal state of a body (i.e., the degree of hotness or coolness of the body) and determines the direction of flow of heat on keeping the two bodies at different temperatures in contact. |
| S.I. unit of heat is Joule (J) | S.I. unit of temperature is Kelvin (K) |
| The amount of heat contained in a body depends on mass, temperature, material of the body | The temperature of a body depends on the average kinetic energy of it's molecules due to their random motion. |
| Heat is measured by the principle of calorimetry. | Temperature is measured by a thermometer. |
| Two bodies having same quantity of heat may differ in their temperature. | Two bodies at same temperature may differ in the quantities of heat contained in them. |
| When two bodies are placed in contact, the total amount of heat is equal to the sum of heat of the individual bodies. | When two bodies at different temperatures are placed in contact, the resultant temperature is a temperature in between the two temperatures . |
What do you understand by thermal expansion of a substance?
Answer
The expansion of a substance on heating is called the thermal expansion of that substance.
Name the three kinds of thermal expansion. Out of solids, liquids and gases, which expands more ?
Answer
The three kinds of thermal expansion are :
- Linear expansion
- Superficial expansion
- Cubical expansion.
On heating, liquids expand more than solids and gases expand much more than liquids. Hence, expansion in gases is the most.
State the volume changes observed when a given mass of water is heated from 0 °C to 10 °C. Sketch a temperature-volume graph to show the behavior.
Answer
The volume of water first decreases on heating it from 0 °C to 4 °C and then increases on further heating it from 4 °C to 10 °C. The volume of water is thus minimum at 4 °C.
Temperature-Volume graph showing the variation in volume of water from 0 °C to 10 °C is shown below:

Draw a graph to show the variation in density of water with temperature in the temperature range from 0 °C to 10 °C.
Answer
The following graph shows the variation in density of water with temperature in the range of 0 °C to 10 °C.

A given mass of water is cooled from 10 °C to 0 °C. State the volume changes observed. Represent these changes on a temperature-volume graph.
Answer
On cooling water from 10 °C, the density of water first increases up to 4 °C and then decreases on cooling further below 4 °C to 0 °C.
Hence, the density of water is maximum at 4 °C which is equal to 1 g cm-3 (or 1000 kg m-3)
Temperature-Volume graph showing these changes is given below:

Explain the following:
Water pipes in colder countries often burst in winter.
Answer
In colder countries, during winter nights, as the temperature starts falling below 4 °C, water in pipe lines expand and it exerts large pressure on the pipes, causing them to burst.
Explain the following:
In winter, water tank (or ocean) starts freezing from the surface and not from the bottom.
Answer
In winter, when the atmospheric temperature drops below 0 °C, the surface water of a tank or ocean initially above 4 °C starts losing heat to the air.
As the surface water temperature falls below 4 °C, it contracts and becomes denser, causing it to sink to the bottom.
This process continues until the entire water mass reaches 4 °C. As the top layers cool below 4 °C, the water expands, reducing its density. Consequently, the water does not sink further but remains at the surface.
When the air temperature falls below 0 °C, the surface water gradually freezes into ice, while the water below the ice layer stays at 4 °C. This is why the freezing of a water tank (or ocean) begins from the top and not the bottom.
Explain the following:
Fishes survive in ponds even when the atmospheric temperature is below 0 °C.
Answer
In winter, when the atmospheric temperature drops below 0 °C, the surface water of a pond cools and contracts. It continues to sink until the entire water mass reaches a temperature of 4 °C.
Further cooling causes the top layers to expand and become less dense, preventing them from sinking. When the air temperature falls below 0 °C, the surface water loses heat and gradually freezes into ice.
However, the water beneath the ice layer remains at 4 °C. The layer of water in contact with the ice is at 0 °C, while the layers below gradually increase in temperature up to 4 °C.
Fish can survive in the pond because the ice acts as an insulator, preventing heat transfer from the water to the atmosphere.
Explain the following:
A hollow glass sphere which floats with its entire volume submerged in water at 4 °C, sinks when water is heated above 4 °C.
Answer
As we know that, the density of water decreases on heating water above 4 °C. Hence, the upthrust acting on the glass sphere decreases and becomes less than the weight of the glass sphere, hence the glass sphere sinks.
Explain the following:
A glass bottle completely filled with water and tightly closed at room temperature, is likely to burst when kept in the freezer of a refrigerator.
Answer
Due to the anomalous nature of water it expands when the temperature goes below 4 °C inside the freezer. As the glass bottle is completely filled with water and tightly closed, so there is no space for the water to expand and hence the bottle bursts.
Describe an experiment to show that water has maximum density at 4 °C. What important consequences follow from this peculiar property of water? Discuss the importance of this phenomenon in nature.
Answer
Hope's apparatus can be used for demonstrating that water has maximum density at 4 °C. The apparatus consists of a tall metallic cylinder with two side openings P, near the top and Q near the bottom, fitted with thermometers T1 and T2 respectively.

The central part of the cylinder is surrounded by a cylindrical trough containing a freezing mixture of ice and salt. The cylinder is filled with pure water at room temperature. The temperature recorded by both the thermometers is observed at regular intervals of time.
Observations —
- Initially both thermometers T1 and T2 show same temperature (i.e., room temperature).
- First the temperature recorded by lower thermometer T2, starts decreasing and finally it becomes steady at 4 °C, while the temperature recorded in upper thermometer T1 remains almost unchanged during this time.
- While the temperature recorded by lower thermometer T2 remains constant at 4 °C, the upper thermometer T1 shows a continuous fall in temperature up to 0 °C and then it also becomes steady.
- At this stage, the lower thermometer T2 shows the temperature 4 °C at which water has the maximum density while the upper thermometer T1 shows the temperature of water and ice at 0 °C.
Important consequences that follow due to this peculiar property of water (i.e., anomalous expansion of water) are:
- It is responsible for bursting of water pipelines and destruction of crops during very cold nights.
- It helps in preserving aquatic life during very cold weather.
In nature, during winter when the atmospheric temperature starts falling below 0 °C, water at the surface of a pond initially at temperature above 4 °C, begins to radiate heat to the atmosphere, so the temperature of water near the surface starts falling.

When temperature of water at the surface falls below 4 °C, water contracts and its density increases and therefore, it sinks to the bottom.
This continues till temperature of entire water reaches to 4 °C. Now, further cooling of top layers below 4 °C results in expansion of water and so its density decreases.
As a result, water does not sink further, but it remains on the surface. When the temperature of atmosphere falls below 0 °C, water on the surface looses further heat to the atmosphere and gradually freezes into ice, but water below the ice layer remains at 4 °C.
The water layer just below the ice in contact with it will be at 0 °C, as shown in figure. Since, ice is a poor conductor of heat, so ice now prevents the flow of heat from water of the pond to the atmosphere.
Thus, temperature of water in contact with ice is at 0 °C and that of layers below the ice gradually increases to 4 °C. As a result, fish and other aquatic creatures remain alive in water of the pond (or lake), though water on the surface has frozen into ice. Nature thus protects the aquatic life during the winter season.
Draw a diagram showing the temperature of various layers of water in an ice covered pond.
Answer
Below diagram shows the temperature of various layers of water in an ice covered pond:

The gases which are good ............... of long wavelength infrared radiations are called greenhouse gases.
- emitters
- reflectors
- absorbers
- none of the above
Answer
absorbers
Reason — Green house effect is the process of warming of the earth's surface and it's lower atmosphere, by absorption of infrared radiations of long wavelength emitted out from the surface of the earth by greenhouse gases such as carbon dioxide, methane, nitrous oxide, ozone, chlorofluorocarbon (CFCs), etc.
Which of the following is a green house gas?
- oxygen
- nitrogen
- chlorine
- carbon dioxide
Answer
carbon dioxide
Reason — Carbon dioxide is a green house gas. Other green house gases are methane, nitrous oxide, ozone and chlorofluorocarbon.
The increase of carbon dioxide gas in atmosphere will cause —
- decrease in temperature
- increase in temperature
- decrease in humidity
- increase in humidity.
Answer
increase in temperature
Reason — The increase of carbon dioxide gas in atmosphere will cause an increase in green house effect hence increasing global warming and increase in temperature.
Without green house effect, the average temperature of earth’s surface would have been —
- -18 °C
- 33 °C
- 0 °C
- 15 °C
Answer
-18 °C
Reason — In the absence of green house gases from the atmosphere, entire heat energy radiated from the earth's surface and objects on it, would escape out into space and then average temperature on earth would be -18 °C instead of 15.5 °C .
Global warming means ............... in average effective temperature near the earth’s surface due to ............... in the amount of greenhouse gases in its atmosphere.
- decrease, increase
- decreases, decrease
- increase, decrease
- increase, increase
Answer
increase, increase
Reason — Global warming means a increase in average effective temperature near the earth's surface due to an increase in the amount of green house gases in its atmosphere.
The global warming has resulted in —
- the increase in yield of crops
- the decrease in sea levels
- the decrease in human deaths
- the increase in sea levels.
Answer
the increase in sea levels
Reason — Due to global warming melting of ice is more and the area of sea ice around the poles is shrinking. It is expected that in the next 50 years, the ice at both the poles will melt completely and therefore the sea levels will rise.
The conversion of part of energy into an unuseful form of energy is called ............... .
Answer
The conversion of part of energy into an unuseful form of energy is called degradation of energy.
What do you mean by greenhouse effect?
Answer
Green house effect is the process of warming of the earth's surface and its lower atmosphere, by absorption of infrared radiations of long wavelength emitted out from the surface of the earth by greenhouse gases such as carbon dioxide, methane, nitrous oxide, ozone, chlorofluorocarbon (CFCs), etc.
Name three greenhouse gases.
Answer
Three green house gases are — carbon dioxide, methane, nitrous oxide.
Which of the following solar radiations pass through the atmosphere of the earth?
X-rays, ultraviolet rays, visible light rays, infrared radiation.
Answer
The solar radiations that pass through the atmosphere of the earth are visible light rays and infrared radiation.
Name the radiations which are absorbed by the greenhouse gases.
Answer
The radiations which are absorbed by the green house gases are infrared radiation of long wavelength.
What would have been the temperature of earth's atmosphere in absence of greenhouse gases in it?
Answer
In the absence of green house gases the average temperature on earth would be -18 °C instead of 15.5 °C.
What results in the increase of carbon dioxide contents of earth’s atmosphere?
Answer
The main human activities responsible for increase of the carbon dioxide gas in the earth's atmosphere are —
- The burning of fuels, deforestation, transportation and industrial production (particularly cement factories).
- Increase of population (human beings emit nearly 32 gigatonnes of carbon dioxide each year).
- Imbalance of carbon dioxide cycle (the ocean does not absorb full amount of carbon dioxide and plants are not able to change all the carbon dioxide into oxygen).
The increase in concentration of carbon dioxide due to the above human activities is responsible for 60% increase in green house effect.
State the effect of greenhouse gases on the temperature of earth’s atmosphere.
Answer
The green house gases have an average warming effect of nearly 33 °C so that the average temperature on earth's surface is about 15.5 °C.
What do you mean by global warming?
Answer
Global warming means the increase in average effective temperature near the earth's surface due to an increase in the amount of green house gases in its atmosphere.
What causes the rise in atmospheric temperature?
Answer
The reason for rise in atmospheric temperature is the increase in amount of green house gases present in the atmosphere.
As the green house gases increase they trap more and more of heat radiated from the earth's surface. Thereby increasing the temperature of the earth.
State the cause of increase of greenhouse effect.
Answer
The reason for the increase in green house effect is the increase in green house gases like carbon dioxide and methane.
Human activities responsible for increase of the carbon dioxide gas in the earth's atmosphere are —
- The burning of fuels, deforestation, transportation and industrial production (particularly cement factories).
- Increase of population (human beings emit nearly 32 gigatonnes of carbon dioxide each year).
- Imbalance of carbon dioxide cycle (the ocean does not absorb full amount of carbon dioxide and plants are not able to change all the carbon dioxide into oxygen).
The increase in concentration of carbon dioxide due to the above human activities is responsible for 60% increase in green house effect.
Apart from this, rice cultivation, animal husbandry, natural gas exploration, burning of bio mass in clearing of forests and leakage in natural gas pipe line have doubled the concentration of methane which is also responsible for the increase in green house effect.
What will be the effect of global warming at the poles?
Answer
Due to global warming, melting of ice is more and the area of sea ice around the poles is shrinking. It is expected that in the next 50 years, the ice at both the poles will melt completely and therefore the sea level will rise.
State the effect of global warming in coastal regions.
Answer
As more ice is melting due to the effect of global waring, so, buildings and roads in the coastal areas will get flooded and they could suffer damage from hurricanes and tropical storms.
How will global warming affect the sea level?
Answer
Due to global warming, melting of ice is more and the area of sea ice around the poles is shrinking. It is expected that in the next 50 years, the ice at both the poles will melt completely and therefore the sea level will rise.
Buildings and roads in the coastal areas will get flooded and they could suffer damage from hurricanes and tropical storms.
How will global warming affect agriculture?
Answer
In the near future, warming of nearly 3 °C will result in poor yield in farms in low latitude regions and will increase the rise of malnutrition. The crop yield is expected to increase in middle and high latitude regions.
Therefore, the farmers will have to shift their farming region from low latitudes to high latitudes.
State two ways to minimize the impact of global warming.
Answer
Ways to minimize the impact of global warming are —
- Use of renewable source of energy for generation of electricity instead of electricity from fossil fuel-based power plants.
- Use of battery-operated vehicles such as electric cars, bikes and scooters.
What is carbon tax? Who will pay it?
Answer
Since, industries emit carbon dioxide to a great extent, to check them, they must be asked to pay carbon tax.
This tax can be calculated on the basis of carbon emission from the industry, number of employee hours and turn over of the industry.
This will encourage the industry to use energy efficient offices and to avoid the travelling of its employees by having tele-conferencing.
State in detail, the causes of global warming.
Answer
The cause of global warming is the increase in concentration of greenhouse gases present in the atmosphere of the earth due to human activities. The increase in different green house is as follows:
- The concentration of carbon dioxide has increased upt 25% due to industrial growth, combustion of fossil fuels, clearing of forests, etc.
- The concentration of methane has doubled due to agricultural sources such as rice cultivation and animal husbandry, natural gas exploration, burning of biomass, clearing of forests and leakage in natural gas pipelines.
- The concentration of chlorofluorocarbons has increased at a rate of 5% per year.
What technological measures are necessary to minimise the impact of global warming ?
Answer
Three technological measures are necessary to minimise the impact of global warming :
- Use of renewable sources of energy for generation of electricity instead of generating electricity from fossil fuel-based power plants — Fossil fuel-based power plants are major contributors to greenhouse gas emissions, accounting for around 21.3% of total emissions. To mitigate this impact, it is essential to shift towards alternative energy sources like wind, solar, tidal, and geothermal energy for electricity generation. Thus, it is necessary to phase out fossil fuel-based power plants and encourage the adoption of cleaner energy sources.
- Use of battery operated vehicles such as electric cars, bikes and scooters — Traditional vehicles powered by internal combustion engines contribute approximately 14% of the total greenhouse gas emission. Switching to battery-operated vehicles and charging them with electricity from renewable sources will greatly decrease carbon dioxide emissions. These vehicles must be used in full capacity and size of the vehicles must be reduced.
- Use of bio-char stoves for cooking — In developing countries, biomass is commonly used for cooking, leading to a significant increase in greenhouse gas emissions. To address this issue, it is crucial to employ new technology that utilizes specially designed bio-char stoves. These stoves burn biomass in the absence of air, resulting in the production of smokeless combustible gases like methane and hydrogen, leaving a charcoal residue which can be buried in soil.
What policy measures should the government enforce in order to minimise global warming ?
Answer
The policy measures that the government should enforce in order to minimise global warming are:
- Educating children to live a sustainable lifestyle — We need to educate the children that genuine happiness lies in a less competitive and more cooperative society. Consuming more and buying more must not be the aim of life. For a sustainable life, we must make full use of what we have. The materialistic gains give only temporary happiness.
- Controlling population through family planning, welfare reforms and empowerment of women — The world population is expected to increase from 7.1 billion in 2012 to 9.15 billion by the year 2050, with most of the growth taking place in developing countries. This needs to be controlled. Following measures can be taken:
- free and easy access to family planning
- welfare provisions to encourage smaller families
- empowerment of women and freedom to choose their career (because educated women are more conscious about family planning due to their career commitments.)
What do you mean by degradation of energy? Explain it by taking two examples of your daily 1ife.
Answer
The gradual decrease of useful energy due to radiation loss, friction, etc. is called degradation of energy.
Examples of degradation of energy —
- In transmission of electricity from a power generating station, a lot of electrical energy is wasted in the form of heat energy in the line wires used for transmission.
- When we cook food over a fire, the major part of heat energy obtained from the fuel is radiated out in the atmosphere. This radiated energy is of no use to us and thus it is a degraded form of energy.
Assertion (A) : Fishes survive in ponds even when the atmospheric temperature is below 0 °C.
Reason (R) : The anomalous expansion of water helps in preserving aquatic life.
- Both A and R are true and R is the correct explanation of A
- Both A and R are true and R is not the correct explanation of A
- Assertion is false but reason is true
- Assertion is true but reason is false
Answer
Both A and R are true and R is the correct explanation of A
Explanation
Assertion (A) is true because this happens in cold regions during winter, even when the air freezes, fish can survive underwater.
Reason (R) is true because of water's anomalous expansion, it is densest at 4 °C, so water at 4 °C stays at the bottom, while water above 4 °C (and eventually ice) remains at the top. This keeps the lower layers liquid and habitable for aquatic life. Hence, the anomalous expansion of water explains why the bottom of the pond remains unfrozen, allowing fish to survive.
A scientific research team records temperature data of a freshwater lake in the Himalayas during winter. Three thermometers A, B, and C are used, where A is calibrated in the Celsius scale, B in the Fahrenheit scale, and C in the Kelvin scale.
On a particular night, when the surface water of the lake freezes, the recorded temperatures are :
A ⟶ 0 °C, B ⟶ 32 °F, C ⟶ 273 K
Next morning, the air temperature increases slightly, but the ice on the surface of the lake does not melt immediately.
(a) What happens to the fish present in the lake when the surface of the lake freezes?
(b) If the temperature of water at the bottom of the lake is measured, what would be the readings on thermometers A, B, and C?
(c) Why does the ice formed on the surface of the lake not sink?
(d) Why does the ice on the lake take time to melt even after the air temperature rises above 0 °C?
(e) Why is the Kelvin scale preferred in scientific calculations related to heat transfer?
Answer
(a) When the surface of the lake freezes, the fish remain safe in the water below because only the upper layer freezes, while the water beneath stays liquid (around 4 °C).
The ice layer acts as an insulating cover, preventing further heat loss, allowing aquatic life to survive.
(b) The water at the bottom of the lake remains at about 4°C because water has maximum density at 4°C.
Thermometer A = 4°C
To convert into Fahrenheit:
F = C + 32
F = × 4 + 32
F = 7.2 + 32 = 39.2°F
Thermometer B = 39.2°F
To convert into Kelvin:
K = °C + 273
K = 4 + 273 = 277 K
Thermometer C = 277 K
(c) Ice does not sink because it is less dense than liquid water. When water freezes, its structure expands, decreasing its density.
(d) Even when the air temperature rises above 0°C, the ice does not melt immediately because it must absorb latent heat of fusion.
This heat is required to change ice into water without raising its temperature. Hence, melting takes place gradually and the ice takes time to melt.
(e) The Kelvin scale is preferred in scientific calculations because it is an absolute scale of temperature starting from absolute zero.
It has no negative values and is directly related to the kinetic energy of molecules. Therefore, it is more convenient and accurate for calculations in heat transfer and thermodynamics.
Two cities, P and Q, have identical population and climate. City P uses coal-based thermal power and diesel vehicles, whereas City Q uses solar and wind energy along with electric vehicles.
(a) Which city is likely to show a higher average temperature rise after 10 years?
(b) Identify the main greenhouse gases responsible for the higher temperature rise in the city mentioned above by you in (a).
(c) "Energy is conserved, so using fossil fuels or renewable sources makes no difference."
Is this statement scientifically correct? Give reason.
(d) Give physics-based reasons to explain which city will have long-term energy sustainability.
Answer
(a) City P is likely to show a higher average temperature rise after 10 years.
This is because coal-based thermal power plants and diesel vehicles release large amounts of greenhouse gases which enhance the greenhouse effect and increase atmospheric temperature.
(b) The main greenhouse gases responsible are:
- Carbon dioxide (CO2)
- Methane (CH4)
- Nitrous oxides (N2O)
(c) No, the statement is not scientifically correct.
Although energy is conserved, the source and form of energy make a great difference. Fossil fuels are exhaustible and release harmful gases and pollutants when used. Renewable sources such as solar and wind energy are naturally replenished and produce much less pollution. Therefore, both do not have the same environmental effect.
(d) City Q has better long-term energy sustainability.
This is because solar and wind energy are renewable and non-exhaustible sources of energy. They depend on natural processes that continue continuously, unlike fossil fuels which are limited in quantity. Also, they cause much less pollution and reduce greenhouse gas emissions, making them more suitable for long-term sustainable use.