The formula of a compound represents
- an atom
- a particle
- a molecule
- a combination
Answer
a molecule
Reason — The formula of a compound employs symbols to denote the molecule of a compound.
An atom is the smallest unit of an element that retains its chemical properties (like H, O, Na). A formula of a compound does not represent a single atom, because compounds are made of two or more different atoms chemically combined. A particle is a very broad term. It includes atom, molecule, ion.<>
The correct formula of aluminium oxide is
- AlO3
- AlO2
- Al2O3
- Al3O2
Answer
Al2O3
Reason — The formula of aluminium oxide can be determined as below:
The valency of lead in lead oxide (PbO) is
- one
- two
- three
- four
Answer
two
Reason — The valency of lead in lead oxide can be determined from the formula PbO as below:
But valency of O is 2. Multiplying by 2, we get:
Therefore, valency of Lead is 2.
Which of the following elements has a molecular mass that is four times its atomic mass?
- Oxygen
- Neon
- Phosphorus
- Sulphur
Answer
Phosphorus
Reason
Atomic mass of P = 31 μ
Molecular form = P4 ⟶ Molecular mass = 31 × 4 = 124 μ
124 μ is 4 times the atomic mass
Hence, phosphorus has molecular mass four times its atomic mass.
Oxygen is a diatomic molecule. Neon exists as single atoms. Sulphur exists as S8 molecules.
Match the following : (Refer common names in the beginning of the book)
| Compound | Formula |
|---|---|
| (a) Boric acid | (i) NaOH |
| (b) Phosphoric acid | (ii) SiO2 |
| (c) Nitrous acid | (iii) Na2CO3 |
| (d) Nitric acid | (iv) KOH |
| (e) Sulphurous acid | (v) CaCO3 |
| (f) Sulphuric acid | (vi) NaHCO3 |
| (g) Hydrochloric acid | (vii) H2S |
| (h) Silica (sand) | (viii) H2O |
| (i) Caustic soda (sodium hydroxide) | (ix) PH3 |
| (j) Caustic potash (potassium hydroxide) | (x) CH4 |
| (k) Washing soda (sodium carbonate) | (xi) NH3 |
| (l) Baking soda (sodium bicarbonate) | (xii) HCl |
| (m) Lime stone (calcium carbonate) | (xiii) H2SO3 |
| (n) Water | (xiv) HNO3 |
| (o) Hydrogen sulphide | (xv) HNO2 |
| (p) Ammonia | (xvi) H3BO3 |
| (q) Phosphine | (xvii) H3PO4 |
| (r) Methane | (xviii) H2SO4 |
Answer
| Compound | Formula |
|---|---|
| (a) Boric acid | (xvi) H3BO3 |
| (b) Phosphoric acid | (xvii) H3PO4 |
| (c) Nitrous acid | (xv) HNO2 |
| (d) Nitric acid | (xiv) HNO3 |
| (e) Sulphurous acid | (xiii) H2SO3 |
| (f) Sulphuric acid | (xviii) H2SO4 |
| (g) Hydrochloric acid | (xii) HCl |
| (h) Silica (sand) | (ii) SiO2 |
| (i) Caustic soda (sodium hydroxide) | (i) NaOH |
| (j) Caustic potash (potassium hydroxide) | (iv) KOH |
| (k) Washing soda (sodium carbonate) | (iii) Na2CO3 |
| (l) Baking soda (sodium bicarbonate) | (vi) NaHCO3 |
| (m) Lime stone (calcium carbonate) | (v) CaCO3 |
| (n) Water | (viii) H2O |
| (o) Hydrogen sulphide | (vii) H2S |
| (p) Ammonia | (xi) NH3 |
| (q) Phosphine | (ix) PH3 |
| (r) Methane | (x) CH4 |
Select the basic and acidic radicals in the following compounds.
(a) MgSO4
(b) (NH4)2SO4
(c) Al2(SO4)3
(d) ZnCO3
(e) Mg(OH)2
Answer
The basic and acidic radicals in the compounds are given in the table below:
| S. No. | Compound | Basic radicals | Acidic radicals |
|---|---|---|---|
| a | MgSO4 | Mg2+ | SO42- |
| b | (NH4)2SO4 | NH4+ | SO42- |
| c | Al2(SO4)3 | Al3+ | SO42- |
| d | ZnCO3 | Zn2+ | CO32- |
| e | Mg(OH)2 | Mg2+ | OH- |
Give the formula and valency of :
(a) aluminate
(b) chromate
(c) aluminium
(d) cupric
Answer
The formula and valency are given in the table below:
| S. No. | Name | Formula | Valency |
|---|---|---|---|
| a | aluminate | AlO33- | 3 |
| b | chromate | CrO42- | 2 |
| c | aluminium | Al | 3 |
| d | cupric | Cu2+ | 2 |
What do the following symbols stand for ?
(a) H
(b) H2
(c) 2H
(d) 2H2
Answer
(a) H → One atom of Hydrogen.
(b) H2 → One molecule of Hydrogen.
(c) 2H → Two atoms of Hydrogen.
(d) 2H2 → Two molecules of Hydrogen.
Write the chemical names of the following compounds :
(a) Ca3(PO4)2
(b) K2CO3
(c) K2MnO4
(d) Mn3(BO3)2
(e) Mg(HCO3)2
(f) Na4Fe(CN)6
(g) Ba(ClO3)2
(h) Ag2SO3
(i) (CH3COO)2Pb
(j) Na2SiO3
Answer
(a) Ca3(PO4)2 → Calcium Phosphate
(b) K2CO3 → Potassium Carbonate
(c) K2MnO4 → Potassium Manganate
(d) Mn3(BO3)2 → Manganese (II) borate
(e) Mg(HCO3)2 → Magnesium Hydrogen Carbonate
(f) Na4Fe(CN)6 → Sodium Ferrocyanide
(g) Ba(ClO3)2 → Barium Chlorate
(h) Ag2SO3 → Silver Sulphite
(i) (CH3COO)2Pb → Lead Acetate
(j) Na2SiO3 → Sodium Silicate
Write chemical formulae of the sulphates of Aluminium, Ammonium and Zinc.
Answer
(a) Chemical formula of Aluminium Sulphate:
∴ Chemical formula of Aluminium Sulphate is Al2(SO4)3
(b) Chemical formula of Ammonium Sulphate:
∴ Chemical formula of Ammonium Sulphate is (NH4)2SO4
(c) Chemical formula of Zinc Sulphate:
∴ Chemical formula of Zinc Sulphate is ZnSO4
The valency of an element A is 3 and that of element B is 2. Write the formula of the compound formed by the combination of A and B.
Answer
Formula of the compound formed by the combination of A and B:
∴ Formula of the compound is A2B3
Give the names of the following compounds.
(a) KClO
(b) KClO2
(c) KClO3
(d) KClO4
Answer
(a) KClO → Potassium hypochlorite
(b) KClO2 → Potassium Chlorite
(c) KClO3 → Potassium Chlorate
(d) KClO4 → Potassium Perchlorate
The formula of the sulphate of an element M is M2(SO4)3. Write the formula of its
(a) Chloride
(b) Oxide
(c) Phosphate
(d) Acetate
Answer
Given, formula of sulphate is M2(SO4)3.
∴ Valency of M is 3.
(a) Formula of Chloride:
∴ Formula of chloride of element M is MCl3
(b) Formula of Oxide:
∴ Formula of oxide of element M is M2O3
(c) Formula of Phosphate:
∴ Formula of phosphate of element M is MPO4
(d) Formula of Acetate:
∴ Formula of acetate of element M is M(CH3COO)3
What is a symbol of an element ? What information does it convey ?
Answer
A symbol is a short form that stands for the atom of a specific element or the abbreviation used for the name of an element.
A symbol conveys the following information:
- It represents the name of the element.
- It represents one atom of the element.
- It represents a definite mass of the element (equal to atomic mass expressed in grams).
Why is the symbol S for sulphur, but Na for sodium and Si for silicon ?
Answer
When the first letter of more than one element is the same the elements are denoted by two letters. Sulphur, Sodium and Silicon all have the first letter as S. Therefore, only sulphur is denoted by S, Silicon is denoted by two letter Si, Sodium is denoted by two letter Na taken from its latin name Natrium.
Write the full form of IUPAC.
Answer
Full form of IUPAC is International Union of Pure and Applied Chemistry.
Name the elements represented by the following symbols :
Au, Pb, Sn, Hg.
Answer
The elements represented by the following symbols are:
Au → Gold
Pb → Lead
Sn → Tin
Hg → Mercury
If the symbol for Cobalt, Co, were written as CO, what would be wrong with it ?
Answer
When writing symbols, we need to be careful about the case of the letters in the symbol. CO means the compound Carbon Monoxide not Cobalt.
Explain the terms 'valency' and 'variable valency'.
Answer
Valency is the combining capacity of an atom or of a radical. The valency of an element or of a radical is the number of hydrogen atoms that will combine with or displace one atom of that element or radical.
Variable valency is the ability of certain elements to have more than one valency or different combining capacities. An atom of an element can sometimes lose more electrons than are present in its valence shell, i.e., there is a loss of electrons from the penultimate shell too. Such element is said to exhibit variable valency.
How are the elements with variable valency named ? Explain with an example.
Answer
If an element exhibits two different positive valencies, then the suffix "ous" is used for the lower valency and the suffix "ic" is used for the higher valency. Modern chemists use Roman numerals in place of these trivial names.
For example, Iron can exhibit a valency of +2 or +3. The ion with valency +2 is named as Ferrous ion (Fe2+) and with valency +3 is named as Ferric ion (Fe3+). Its oxides will be named as Ferrous oxide or Iron (II) oxide [FeO] and Ferric oxide or Iron (III) oxide [Fe2O3].
(a) What is a chemical formula ?
(b) What is the significance of a formula ? Give an example to illustrate.
Answer
(a) A chemical formula also known as molecular formula employs symbols to denote the molecule of an element or of a compound.
(b) The chemical/molecular formula of a compound has quantitative significance. It represents:
- both the molecule and the molecular mass of the compound.
- the respective numbers of different atoms present in one molecule of the compound.
- the ratios of the respective masses of the elements present in the compound.
For example, the formula CO2 means that:
- the molecular formula of carbon dioxide is CO2.
- each molecule contains one carbon atom joined by chemical bonds with two oxygen atoms.
- the molecular mass of carbon dioxide is 44, given that the atomic mass of carbon is 12 and that of oxygen is 16.
What do you understand by the following terms ?
(a) Acid radical
(b) Basic radical
Answer
(a) An Acid radical is the radical that remains after an acidic molecule loses a hydrogen ion (H+). Acid radicals typically have a negative charge. They are also called electronegative radicals or anions.
(b) A Basic radical is the radical that remains after a base molecule loses a hydroxyl ion (OH-). Basic radicals typically have a positive charge. They are also called electropositive radicals or cations.
Write the basic radicals and acidic radicals of the following and then write the chemical formulae of these compounds.
(a) Barium sulphate
(b) Bismuth nitrate
(c) Calcium bromide
(d) Ferrous sulphide
(e) Chromium sulphate
(f) Calcium silicate
(g) Stannic oxide
(h) Sodium zincate
(i) Magnesium phosphate
(j) Sodium thiosulphate
(k) Stannic phosphate
(l) Nickel bisulphate
(m) Potassium manganate
(n) Potassium ferrocyanide
Answer
| S. No. | Compound | Basic radical | Acidic radical | Formula |
|---|---|---|---|---|
| a | Barium sulphate | Ba2+ | SO42- | BaSO4 |
| b | Bismuth nitrate | Bi3+ | NO3- | Bi(NO3)3 |
| c | Calcium bromide | Ca2+ | Br- | CaBr2 |
| d | Ferrous sulphide | Fe2+ | S2- | FeS |
| e | Chromium sulphate | Cr3+ | SO42- | Cr2(SO4)3 |
| f | Calcium silicate | Ca2+ | SiO32- | CaSiO3 |
| g | Stannic oxide | Sn4+ | O2- | SnO2 |
| h | Sodium zincate | Na1+ | ZnO22- | Na2ZnO2 |
| i | Magnesium phosphate | Mg2+ | PO43- | Mg3(PO4)2 |
| j | Sodium thiosulphate | Na1+ | S2O32- | Na2S2O3 |
| k | Stannic phosphate | Sn4+ | PO43- | Sn3(PO4)4 |
| l | Nickel bisulphate | Ni2+ | HSO4- | Ni(HSO4)2 |
| m | Potassium manganate | K1+ | MnO42- | K2MnO4 |
| n | Potassium ferrocyanide | K1+ | Fe(CN)64- | K4[Fe(CN)6] |
Give the names of the elements and number of atoms of those elements, present in the following compounds.
(a) Sodium sulphate
(b) Quick lime
(c) Baking soda
(d) Ammonia
(e) Ammonium dichromate
Answer
(a) Sodium sulphate
Formula: Na2SO4
∴ It contains 2 atoms of Sodium (Na), 1 atom of Sulphur (S) and 4 atoms of Oxygen (O).
(b) Quick lime
Formula: CaO
∴ It contains 1 atom of Calcium (Ca) and 1 atom of Oxygen (O).
(c) Baking soda
Formula: NaHCO3
∴ It contains 1 atom of Sodium (Na), 1 atom of Hydrogen (H), 1 atom of Carbon (C) and 3 atoms of Oxygen (O).
(d) Ammonia
Formula: NH3
∴ It contains 1 atom of Nitrogen (N) and 3 atoms of Hydrogen (H).
(e) Ammonium dichromate
Formula: (NH4)2Cr2O7
∴ It contains 2 atoms of Nitrogen (N), 8 atoms of Hydrogen (H), 2 atoms of Chromium (Cr) and 7 atoms of Oxygen (O).
The valency of an element A is 4. Write the formula of its:
(a) oxide
(b) nitrate
(c) phosphate
Answer
(a) Chemical formula of Oxide of A:
∴ Chemical formula of Oxide of A is AO2
(b) Chemical formula of Nitrate of A:
∴ Chemical formula of Nitrate of A is A(NO3)4
(c) Chemical formula of Phosphate of A:
∴ Chemical formula of Phosphate of A is A3(PO4)4
Identify the cations (electropositive ions) and anions (electronegative ions) in the following compounds:
(a) CH3COONa
(b) NH4Cl
(c) PbCl2
(d) MgO
Answer
(a) CH3COONa (Sodium acetate)
Cation: Na+ (Sodium ion)
Anion: CH3COO- (Acetate ion)
(b) NH4Cl (Ammonium chloride)
Cation: NH4+ (Ammonium ion)
Anion: Cl- (Chloride ion)
(c) PbCl2 (Lead(II) chloride)
Cation: Pb2+ (Lead(II) ion)
Anion: Cl- (Chloride ion)
(d) MgO (Magnesium oxide)
Cation: Mg2+ (Magnesium ion)
Anion: O2- (Oxide ion)
Depending on the number of oxygen atoms in the anions, the names of some acids and their formulae are given. Write the formula of their corresponding salts.
| Acid | Formula | Salt | Formula |
|---|---|---|---|
| Perchloric acid | HClO4 | Sodium perchlorate | NaClO4 |
| Chloric acid | HClO3 | Sodium chlorate | ............... |
| Chlorous acid | HClO2 | Potassium chlorite | ............... |
| Hypochlorous acid | HClO | Sodium hypochlorite | ............... |
Answer
| Acid | Formula | Salt | Formula |
|---|---|---|---|
| Perchloric acid | HClO4 | Sodium perchlorate | NaClO4 |
| Chloric acid | HClO3 | Sodium chlorate | NaClO3 |
| Chlorous acid | HClO2 | Potassium chlorite | KClO2 |
| Hypochlorous acid | HClO | Sodium hypochlorite | NaClO |
Give an example of a divalent anion and a trivalent cation. Write the formula of the compound formed by their combination.
Answer
Aluminium is trivalent cation with a valency of 3 → Al3+
Sulphate is divalent anion with a valency of 2 → SO42-
Formula:
Hence, the formula is : Al2(SO4)3
Write the Latin names of sodium, mercury and iron.
Answer
| Name | Latin Name |
|---|---|
| Sodium | Natrium |
| Mercury | Hydrargyrum |
| Iron | Ferrum |
State the number of atoms present in each of the following:
(a) PO43-
(b) P2O5
(c) H2SO4
(d) SO4-2
Answer
(a) PO43-
P = 1 atom
O = 4 atoms
Total atoms = 1 + 4 = 5 atoms
(b) P2O5
P = 2 atoms
O = 5 atoms
Total atoms = 2 + 5 = 7 atoms
(c) H2SO4
H = 2 atoms
S = 1 atom
O = 4 atoms
Total atoms = 2 + 1 + 4 = 7 atoms
(d) SO4-2
S = 1 atom
O = 4 atoms
Total atoms = 1 + 4 = 5 atoms
What is a chemical equation ? Why it is necessary to balance it ?
Answer
A chemical equation is the symbolic representation of a chemical reaction using the symbols and formulae of the substances involved in the reaction.
An equation must be balanced in order to comply with the "Law of Conservation of Matter", which states that matter is neither created nor destroyed in the course of a chemical reaction. An unbalanced equation would imply that atoms have been created or destroyed.
State the information conveyed by the following equation.
Zn(s) + 2HCl (aq) ⟶ ZnCl2 (aq) + H2↑
Answer
The given equation tells us:
- About the reactants involved and the products formed as a result of the reaction and their state. Zinc (Zn) and Hydrochloric acid (HCl) are the reactants. Zinc is in solid state and HCl is in aqueous solution state. The product Zinc Chloride (ZnCl2) is also in aqueous solution state and Hydrogen is in gaseous state.
- About the number of molecules of each substance taking part and formed in the reaction. Here one molecule of Zinc and two molecules of Hydrochloric acid react to give one molecule of Zinc chloride and one molecule of Hydrogen gas.
- About chemical composition of respective molecules. For example, one molecule of Zinc chloride contains one atom of Zinc and two atoms of Chlorine.
- About molecular mass; that 65 parts by weight of Zinc reacts with 73 parts by weight of Hydrochloric acid to produce 136 parts by weight of Zinc chloride and 2 parts by weight of Hydrogen.
- 65 g of Zinc on treatment with 73 g of HCl, will produce 22.4 litres of hydrogen gas at S.T.P.
- It also proves the law of conservation of mass. According to the above equation, 138 gram of reactants are producing 138 gram of products.
What is the limitation of the reaction given in question 2?
Answer
The equation of the given reaction does not tell us:
- the time taken for the completion of the reaction.
- whether heat is given out or absorbed during the reaction.
- the respective concentrations of the reactants and the products.
- the rate at which the reaction proceeds.
- whether the reaction is completed or it is not completed.
Write chemical equations for the following word equations and balance them.
(a) Carbon + Oxygen ⟶ Carbon dioxide
(b) Nitrogen + Oxygen ⟶ Nitrogen monoxide
(c) Calcium + Nitrogen ⟶ Calcium nitride
(d) Calcium oxide + Carbon dioxide ⟶ Calcium carbonate
(e) Magnesium + Sulphuric acid ⟶ Magnesium sulphate + Hydrogen
(f) Sodium reacts with water to form sodium hydroxide and hydrogen.
Answer
The balanced chemical equations for the word equations are given below:
(a) C + O2 ⟶ CO2
(b) N2 + O2 ⟶ 2NO
(c) 3Ca + N2 ⟶ Ca3N2
(d) CaO + CO2 ⟶ CaCO3
(e) Mg + H2SO4 ⟶ MgSO4 + H2↑
(f) 2Na + 2H2O ⟶ 2NaOH + H2↑
Balance the following equations:
(a) Fe + H2O ⟶ Fe3O4 + H2
(b) Ca + N2 ⟶ Ca3N2
(c) Zn + KOH ⟶ K2ZnO2 + H2
(d) Fe2O3 + CO ⟶ Fe + CO2
(e) PbO + NH3 ⟶ Pb + H2O + N2
(f) Pb3O4 ⟶ PbO + O2
(g) PbS + O2 ⟶ PbO + SO2
(h) S + H2SO4 ⟶ SO2 + H2O
(i) S + HNO3 ⟶ H2SO4 + NO2 + H2O
(j) MnO2 + HCl ⟶ MnCl2 + H2O + Cl2
(k) C + H2SO4 ⟶ CO2 + H2O + SO2
(l) KOH + Cl2 ⟶ KCl + KClO + H2O
(m) NO2 + H2O ⟶ HNO2 + HNO3
(n) Pb3O4 + HCl ⟶ PbCl2 + H2O + Cl2
(o) H2O + Cl2 ⟶ HCl + O2
(p) NaHCO3 ⟶ Na2CO3 + H2O + CO2
(q) HNO3 + H2S ⟶ NO2 + H2O + S
(r) P + HNO3 ⟶ NO2 + H2O + H3PO4
(s) Zn + HNO3 ⟶ Zn(NO3)2 + H2O + NO2
Answer
(a) 3Fe + 4H2O ⟶ Fe3O4 + 4H2
(b) 3Ca + N2 ⟶ Ca3N2
(c) Zn + 2KOH ⟶ K2ZnO2 + H2
(d) Fe2O3 + 3CO ⟶ 2Fe + 3CO2
(e) 3PbO + 2NH3 ⟶ 3Pb + 3H2O + N2
(f) 2Pb3O4 ⟶ 6PbO + O2
(g) 2PbS + 3O2 ⟶ 2PbO + 2SO2
(h) S + 2H2SO4 ⟶ 3SO2 + 2H2O
(i) S + 6HNO3 ⟶ H2SO4 + 6NO2 + 2H2O
(j) MnO2 + 4HCl ⟶ MnCl2 + 2H2O + Cl2
(k) C + 2H2SO4 ⟶ CO2 + 2H2O + 2SO2
(l) 2KOH + Cl2 ⟶ KCl + KClO + H2O
(m) 2NO2 + H2O ⟶ HNO2 + HNO3
(n) Pb3O4 + 8HCl ⟶ 3PbCl2 + 4H2O + Cl2
(o) 2H2O + 2Cl2 ⟶ 4HCl + O2
(p) 2NaHCO3 ⟶ Na2CO3 + H2O + CO2
(q) 2HNO3 + H2S ⟶ 2NO2 + 2H2O + S
(r) P + 5HNO3 ⟶ 5NO2 + H2O + H3PO4
(s) Zn + 4HNO3 ⟶ Zn(NO3)2 + 2H2O + 2NO2
Modern atomic symbols are based on the method proposed by
- Bohr
- Dalton
- Berzelius
- Alchemist
Answer
Berzelius
Reason — Berzelius suggested that the initial letter of an element written in capitals should represent that particular element. This method suggested by him laid the basis of the IUPAC system of chemical symbols and formulae.
The number of carbon atoms in a hydrogen carbonate radical is
- one
- two
- three
- four
Answer
one
Reason — Hydrogen carbonate radical is represented as HCO3-. Thus, it shows that one carbon atom is present in a hydrogen carbonate radical.
The formula of iron (III) sulphate is
- Fe3SO4
- Fe(SO4)3
- Fe2(SO4)3
- FeSO4
Answer
Fe2(SO4)3
Reason — Chemical formula of iron (III) sulphate:
∴ Chemical formula of iron (III) sulphate is Fe2(SO4)3
In water, the hydrogen-to-oxygen mass ratio is
- 1 : 8
- 1 : 16
- 1 : 32
- 1 : 64
Answer
1 : 8
Reason — Hydrogen-to-oxygen mass ratio in water can be found as below:
Molecualr formula of water = H2O
∴ Hydrogen-to-oxygen mass ratio in water
= =
= 1 : 8
The formula of sodium carbonate is Na2CO3 and that of calcium hydrogen carbonate is
- CaHCO3
- Ca(HCO3)2
- Ca2HCO3
- Ca(HCO3)3
Answer
Ca(HCO3)2
Reason — Chemical formula of calcium hydrogen carbonate:
∴ Chemical formula of calcium hydrogen carbonate is Ca(HCO3)2
The correct atomic symbols for carbon, calcium, copper and cadmium respectively are:
- Ca, C, Cu, Cd
- Ca, C, CO, Cd
- C, Ca, Cu, Cd
- Ca, Cl, Co, Cd
Answer
C, Ca, Cu, Cd
Reason — Carbon is denoted by the first letter of its name C, Calcium by the first two letters of its name Ca, Cadmium by first and third letters Cd and Copper is denoted by the first two letters of its latin name Cuprum so Cu.
Hydrargyrum is the Latin name of:
- Gold
- Silver
- Mercury
- Platinum
Answer
Mercury
Reason — The term "Hydrargyrum" is derived from the Greek words "hydr-" meaning water and "argyros" meaning silver. In Latin, "Hydrargyrum" is used to refer to the element mercury, which is a silvery, liquid metal.
In Zn3(PO4)2, the valency of Zn and Phosphate respectively is:
- 3 and 2
- 2 and 6
- 4 and 6
- 2 and 3
Answer
2 and 3
Reason — Given the formula Zn3(PO4)2 :
∴ Valency of Zinc is 2 and Phosphate is 3.
In Ca3(PO4)x, the value of x is:
- 1
- 2
- 3
- 4
Answer
2
Reason — Chemical formula of calcium phosphate:
∴ Chemical formula of calcium phosphate is Ca3(PO4)2 hence x = 2.
In C2H4, the valency of carbon is:
- 2
- 1
- 4
- 3
Answer
4
Reason — Given the formula C2H4 :
∴ Valency of Carbon is 4.
Which of the following represent the correct chemical formula of a compound :
- NaSO4
- AlSO4
- NH4PO4
- Ca3N2
Answer
Ca3N2
Reason — Calcium (Ca) forms a +2 ion (Ca+2) and nitride (N-3) has a –3 charge.
It is perfectly balanced. Therefore, Ca3N2 is correct.
A chemical equation is always balanced to satisfy the conditions of :
Law of constant proportions.
Gay Lussac's law
Law of conservation of mass
Law of mass action.
Answer
Law of conservation of mass
Reason — Law of conservation of mass states that the total mass of the substances on either side of the equation is the same.
In Law of constant proportions, a compound always contains the same elements in a fixed ratio by mass (e.g., water always has hydrogen and oxygen in a fixed ratio). Gay Lussac's law deals with volumes of gases in chemical reactions (gases combine in simple whole-number ratios by volume). Law of mass action relates to the rate of a chemical reaction and how it depends on the concentration of reactants.
One "μ" stands for :
- One atom of any element
- 1/12th of hydrogen atom
- An atom of carbon (C-12)
- 1/12th the mass of carbon atom (C-12).
Answer
1/12th the mass of carbon atom (C-12)
Reason — Atomic mass is expressed in atomic mass units [a.m.u.] or "μ". Atomic mass unit is defined as 1/12 the mass of carbon atom C-12. (The mass of an atom of carbon-12 isotope was given the atomic mass of 12 units, i.e., 12 amu or simply 12 μ ).
Which of the following statements about a compound is incorrect ?
- A molecule of a compound has atoms of different elements.
- A compound cannot be separated into its constituent elements by physical methods of separation.
- A compound retains the physical properties of its constituent elements.
- The ratio of atoms of different elements in a compound is fixed.
Answer
A compound retains the physical properties of its constituent elements.
Reason — When elements combine to form a compound, the resulting substance has completely different physical and chemical properties from the elements that make it up.
A compound is formed by the chemical combination of two or more different elements. The elements in a compound are chemically bonded, so you need chemical methods (like electrolysis) to separate them and not physical ones like filtration or evaporation. Compounds always have a definite composition.
Which of the following reactions is not correct according to the laws?
P4 + 5O2 ⟶ P4O10
C2H6 ⟶ CO2 + H2O
2Zn + O2 ⟶ 2ZnO
CH4 + 2O2 ⟶ CO2 + 2H2O
Answer
C2H6 ⟶ CO2 + H2O
Reason — C2H6 ⟶ CO2 + H2O
This equation does not follow the Law of conservation of mass because reactant side has 2 carbon atoms and 6 hydrogen atoms whereas product side has only 1 carbon atom and 2 hydrogen atoms. Since both the sides do not have the same number of atoms, it violates the Law of conservation of mass which states that matter can neither be created nor destroyed in a chemical reaction.
Other reactions are balanced. The total number of atoms of each element on the reactants side, is the same as the number of atoms in the products formed, in the equation. So, they follow the Law of conservation of mass.
In the compound magnesium nitride (Mg3N2):
P — Mg is trivalent, N is divalent.
Q — Mg is divalent. N is trivalent.
R — Mg is divalent, N is divalent.
Which of the statements given above is/are true?
Only P
Only Q
Only R
Both P and Q
Answer
Only Q
Reason — The valency of elements in compound magnesium nitride (Mg3N2) is,
Hence, Mg is divalent and N is trivalent.
Copper shows variable valency and forms two different compounds with oxygen — Cu2O and CuO.
P — A is cuprous oxide, B is cupric oxide.
Q — A is cupric oxide, B is cuprous oxide.
R — A is copper (I) oxide, B is copper (II) oxide.
Only P
Only Q
Only R
Both P and R
Answer
Both P and R
Reason — Certain elements exhibit more than one valency and they show variable valency.
For example, copper shows valency of 1 and 2 and forms compounds like, cuprous [copper(I)] oxide and cupric [copper(II)] oxide, respectively.
Hence, both P and R are true.
The figure given below shows the molecule of an element, where 'a' denotes the atom with atomic mass 32.
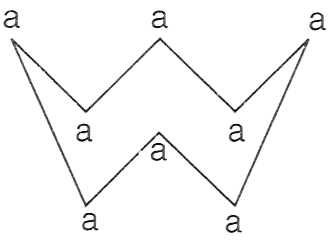
P — Element is tetratomic with molecular mass 128.
Q — Element is octatomic with molecular mass 256.
R — Element is crown-shaped with molecular mass 256.
Only P
Only Q
Only R
Both P and R
Answer
Only Q
Reason — Given element 'a' is having atomic mass 32. The figure shows there are eight 'a' atoms forming a octatomic molecule. So when we calculate the molecular mass it will be
32 x 8 = 256
Hence, element 'a' is octatomic molecule with molecular mass 256.
Fill in the blanks:
(a) Dalton used symbol ............... for oxygen and ............... for hydrogen.
(b) Symbol represents ............... atom of an element.
(c) Symbolic expression for a molecule is called ............... .
(d) Sodium chloride has two radicals. Sodium is a ............... radical while chloride is a ............... radical.
(e) Valency of phosphorus in PCl3 is ............... and in PCl5 is ............... .
(f) Valency of Iron in FeCl2 is ............... and in FeCl3 it is ............... .
(g) Formula of iron (III) carbonate is ............... .
Answer
(a) Dalton used symbol for oxygen and for hydrogen.
(b) Symbol represents one atom of an element.
(c) Symbolic expression for a molecule is called molecular formula .
(d) Sodium chloride has two radicals. Sodium is a basic radical while chloride is a acidic radical.
(e) Valency of phosphorus in PCl3 is 3 and in PCl5 is 5 .
(f) Valency of Iron in FeCl2 is 2 and in FeCl3 it is 3 .
(g) Formula of iron (III) carbonate is Fe2(CO3)3 .
Name the following compounds and state how many atoms of each kind are present in one molecule of these compounds.
(a) H2SO4
(b) HClO4
(c) K2Cr2O7
(d) KMnO4
(e) K4[Fe(CN)6]
(f) Na2CrO4
(g) Mn3(BO4)2
(h) HNO2
Answer
(a) Atoms in H2SO4 are
H ⟶ 2
S ⟶ 1
O ⟶ 4
(b) Atoms in HClO4
H ⟶ 1
Cl ⟶ 1
O ⟶ 4
(c) Atoms in K2Cr2O7
K ⟶ 2
Cr ⟶ 2
O ⟶ 7
(d) Atoms in KMnO4
K ⟶ 1
Mn ⟶ 1
O ⟶ 4
(e) Atoms in K4[Fe(CN)6]
K ⟶ 4
Fe ⟶ 1
C ⟶ 6
N ⟶ 6
(f) Atoms in Na2CrO4
Na ⟶ 2
Cr ⟶ 1
O ⟶ 4
(g) Atoms in Mn3(BO4)2
Mn ⟶ 3
B ⟶ 2
O ⟶ 8
(h) Atoms in HNO2
H ⟶ 1
N ⟶ 1
O ⟶ 2
Correct the following statements.
(a) A molecular formula represents an element.
(b) Molecular formula of water is H2O2.
(c) A molecule of sulphur is monoatomic.
(d) CO and Co both represent cobalt.
(e) Formula of iron (III) oxide is FeO.
Answer
(a) A molecular formula represents the molecule of an element or of a compound.
(b) Molecular formula of water is H2O.
(c) A molecule of sulphur is octatomic.
(d) CO represents Carbon Monoxide and Co represents cobalt.
(e) Formula of iron (III) oxide is Fe2O3.
Give the empirical formula of :
- Benzene (C6H6)
- Glucose (C6H12O6)
- Acetylene (C2H2)
- Acetic acid (CH3COOH)
Answer
- Ratio of C and H atoms in Benzene (C6H6) = 6 : 6
Simplest Ratio = 1 : 1
∴ Empirical formula of Benzene (C6H6) = CH - Ratio of C, H and O atoms in Glucose (C6H12O6) = 6 : 12 : 6
Simplest Ratio = 1 : 2 : 1
∴ Empirical formula of Glucose (C6H12O6) = CH2O - Ratio of C and H atoms in Acetylene (C2H2) = 2 : 2
Simplest Ratio = 1 : 1
∴ Empirical formula of Acetylene (C2H2) = CH - Ratio of C, H and O atoms in Acetic acid (CH3COOH) = 2 : 4 : 2
Simplest Ratio = 1 : 2 : 1
∴ Empirical formula of Acetic acid (CH3COOH) = CH2O
Complete the following table.
| Acid Radicals → Basic Radicals ↓ | Chloride | Nitrate | Sulphate | Carbonate | Hydroxide | Phosphate |
|---|---|---|---|---|---|---|
| Magnesium | MgCl2 | Mg(NO3)2 | MgSO4 | MgCO3 | Mg(OH)2 | Mg3(PO4)2 |
| Sodium | ||||||
| Zinc | ||||||
| Silver | ||||||
| Ammonium | ||||||
| Calcium | ||||||
| Iron (II) | ||||||
| Potassium |
Answer
The completed table is given below:
| Acid Radicals → Basic Radicals ↓ | Chloride | Nitrate | Sulphate | Carbonate | Hydroxide | Phosphate |
|---|---|---|---|---|---|---|
| Magnesium | MgCl2 | Mg(NO3)2 | MgSO4 | MgCO3 | Mg(OH)2 | Mg3(PO4)2 |
| Sodium | NaCl | NaNO3 | Na2SO4 | Na2CO3 | NaOH | Na3PO4 |
| Zinc | ZnCl2 | Zn(NO3)2 | ZnSO4 | ZnCO3 | Zn(OH)2 | Zn3(PO4)2 |
| Silver | AgCl | AgNO3 | Ag2SO4 | Ag2CO3 | AgOH | Ag3PO4 |
| Ammonium | NH4Cl | NH4NO3 | (NH4)2SO4 | (NH4)2CO3 | NH4OH | (NH4)3PO4 |
| Calcium | CaCl2 | CaNO3 | CaSO4 | CaCO3 | Ca(OH)2 | Ca3(PO4)2 |
| Iron (II) | FeCl2 | Fe(NO3)2 | FeSO4 | FeCO3 | Fe(OH)2 | Fe3(PO4)2 |
| Potassium | KCl | KNO3 | K2SO4 | K2CO3 | KOH | K3PO4 |
What is the valency of :
(a) fluorine in CaF2
(b) sulphur in SF6
(c) phosphorus in PH3
(d) carbon in CH4
(e) Manganese in MnO2
(f) Copper in Cu2O
(g) Magnesium in Mg3N2
(h) nitrogen in the following compounds :
(i) N2O3 (ii) N2O5 (iii) NO2 (iv) NO
Answer
(a) Valency of fluorine in CaF2 is -1.
(b) Valency of sulphur in SF6 is +6.
(c) Valency of phosphorus in PH3 is +3.
(d) Valency of carbon in CH4 is +4.
(e) Valency of manganese in MnO2 is +4.
(f) Valency of copper in Cu2O is +1.
(g) Valency of magnesium in Mg3N2 is +2.
(h) Valency of nitrogen in the given compounds :
- N2O3 = +3
- N2O5 = +5
- NO2 = +4
- NO = +2
Why should an equation be balanced ? Explain with the help of a simple equation.
Answer
An equation must be balanced in order to comply with the "Law of Conservation of Matter", which states that matter is neither created nor destroyed in the course of a chemical reaction. An unbalanced equation would imply that atoms have been created or destroyed.
For example, consider the following unbalanced equation:
KNO3 ⟶ KNO2 + O2
In this equation, there are 3 oxygen atoms on the left side, but 4 oxygen atoms on the right side. This means that the equation is not balanced and does not satisfy the law of conservation of mass.
The balanced form of the equation is:
2KNO3 ⟶ 2KNO2 + O2
Now there are 6 oxygen atoms on both the sides. This means that the law of conservation of mass is satisfied and the equation correctly represents the reaction.
Define Atomic mass unit.
Answer
Atomic mass unit is defined as 1⁄12 the mass of an carbon atom C-12.
Define Basic radical.
Answer
A Basic radical is the radical that remains after a base molecule loses a hydroxyl ion (OH-). Basic radicals typically have a positive charge. They are also called electropositive radicals or cations.
Balance the following equations.
(a) Al2O3 + H2SO4 ⟶ Al2(SO4)3 + H2O
(b) NH3 + Cl2 ⟶ NH4Cl + N2
(c) C5H12 (pentane) + O2 ⟶ CO2 + H2O
(d) C4H10 (butane) + O2 ⟶ CO2 + H2O
(e) FeSO4 + H2SO4 + Cl2 ⟶ Fe2(SO4)3 + HCl
(f) NaCl + MnO2 + H2SO4 ⟶ NaHSO4+ MnSO4 + H2O + Cl2
(g) Na2Cr2O7 + HCl ⟶ NaCl + CrCl3 + H2O + Cl2
(h) KMnO4 + H2SO4 + H2S ⟶ K2SO4 + MnSO4 + H2O + S
Answer
(a) Al2O3 + 3H2SO4 ⟶ Al2(SO4)3 + 3H2O
(b) 8NH3 + 3Cl2 ⟶ 6NH4Cl + N2
(c) C5H12 (pentane) + 8O2 ⟶ 5CO2 + 6H2O
(d) 2C4H10 (butane) + 13O2 ⟶ 8CO2 + 10H2O
(e) 2FeSO4 + H2SO4 + Cl2 ⟶ Fe2(SO4)3 + 2HCl
(f) 2NaCl + MnO2 + 3H2SO4 ⟶ 2NaHSO4 + MnSO4 + 2H2O + Cl2
(g) Na2Cr2O7 + 14HCl ⟶ 2NaCl + 2CrCl3 + 7H2O + 3Cl2
(h) 2KMnO4 + 3H2SO4 + 5H2S ⟶ K2SO4 + 2MnSO4 + 8H2O + 5S
Atomic mass of calcium is 40 μ. What does it signify ?
Answer
The atomic mass of calcium being 40 amu signifies that, on average, a calcium atom weighs 40 times the mass of one twelfth of a carbon-12 atom.
Write the balanced chemical equations of the following word equations.
(a) Sodium hydroxide + sulphuric acid ⟶ sodium sulphate + water
(b) Potassium bicarbonate + sulphuric acid ⟶ potassium sulphate + carbon dioxide + water
(c) Iron + sulphuric acid ⟶ ferrous sulphate + hydrogen
(d) Chlorine + sulphur dioxide + water ⟶ sulphuric acid + hydrogen chloride
(e) Silver nitrate ⟶ silver + nitrogen dioxide + oxygen
(f) Copper + nitric acid ⟶ copper nitrate + nitric oxide + water
(g) Ammonia + oxygen ⟶ nitric oxide + water
(h) Barium chloride + sulphuric acid ⟶ barium sulphate + hydrochloric acid
(i) Zinc sulphide + oxygen ⟶ zinc oxide + sulphur dioxide
(j) Aluminium carbide + water ⟶ aluminium hydroxide + methane
(k) Iron pyrites (FeS2) + oxygen ⟶ ferric oxide + sulphur dioxide
(l) Potassium permanganate + hydrochloric acid ⟶ potassium chloride + manganese chloride + chlorine + water
(m) Aluminium sulphate + sodium hydroxide ⟶ sodium sulphate + sodium meta aluminate + water
(n) Aluminium + sodium hydroxide + water ⟶ sodium meta aluminate + hydrogen
(o) Potassium dichromate + sulphuric acid ⟶ potassium sulphate + chromium sulphate + water + oxygen
(p) Potassium dichromate + hydrochloric acid ⟶ potassium chloride + chromium chloride + water + chlorine
(q) Sulphur + nitric acid ⟶ sulphuric acid + nitrogen dioxide + water
(r) Sodium chloride + manganese dioxide + sulphuric acid ⟶ sodium hydrogen sulphate + manganese sulphate + water + chlorine
Answer
(a) 2NaOH + H2SO4 ⟶ Na2SO4 + 2H2O
(b) 2KHCO3 + H2SO4 ⟶ K2SO4 + 2CO2 + 2H2O
(c) Fe + H2SO4 ⟶ FeSO4 + H2
(d) Cl2 + SO2 + 2H2O ⟶ H2SO4 + 2HCl
(e) 2AgNO3 ⟶ 2Ag + 2NO2 + O2
(f) 3Cu + 8HNO3 ⟶ 3Cu(NO3)2 + 2NO + 4H2O
(g) 4NH3 + 5O2 ⟶ 4NO + 6H2O
(h) BaCl2 + H2SO4 ⟶ BaSO4 + 2HCl
(i) 2ZnS + 3O2 ⟶ 2ZnO + 2SO2
(j) Al4C3 + 12H2O ⟶ 4Al(OH)3 + 3CH4
(k) 4FeS2 + 11O2 ⟶ 2Fe2O3 + 8SO2
(l) 2KMnO4 + 16HCl ⟶ 2KCl + 2MnCl2 + 5Cl2 + 8H2O
(m) Al2(SO4)3 + 8NaOH ⟶ 3Na2SO4 + 2NaAlO2 + 4H2O
(n) 2Al + 2NaOH + 2H2O ⟶ 2NaAlO2 + 3H2
(o) 2K2Cr2O7 + 8H2SO4 ⟶ 2K2SO4 + 2Cr2(SO4)3 + 8H2O + 3O2
(p) K2Cr2O7 + 14HCl ⟶ 2KCl + 2CrCl3 + 7H2O + 3Cl2
(q) S + 6HNO3 ⟶ H2SO4 + 6NO2 + 2H2O
(r) 2NaCl + MnO2 + 3H2SO4 ⟶ 2NaHSO4 + MnSO4 + 2H2O + Cl2
Sodium chloride reacts with silver nitrate to produce silver chloride and sodium nitrate
(a) Write the equation.
(b) Check whether it is balanced, if not balance it.
(c) Find the weights of reactants and products.
(d) State the law that this equation satisfies.
Answer
(a) NaCl + AgNO3 ⟶ AgCl + NaNO3
(b) The equation is balanced.
(c) Calculating the weights of reactants and products from the equation:
Thus, weights of reactants = weights of products = 228.5 g
(d) The equation satisfies the law of conservation of mass. It states that the total mass of the substances on either side of the equation is the same.
What information does the following chemical equations convey ?
(a) Zn + H2SO4 ⟶ ZnSO4 + H2
(b) Mg + 2HCl ⟶ MgCl2 + H2
Answer
The above balanced equation conveys the following information:
- One molecule of zinc reacts with one molecule of sulphuric acid to produce one molecule of zinc Sulphate and one molecule of hydrogen.
- 65 g of zinc reacts with 98 g of sulphuric acid to produce 161 g of zinc Sulphate and 2 g of hydrogen.
- 65 g of zinc reacts with 98 g of sulphuric acid to produce 22.4 litres of hydrogen gas at S.T.P.
- It also proves the law of conservation of mass. According to the above equation, 163 gram of reactants are producing 163 gram of products.
The above balanced equation conveys the following information:
- One molecule of magnesium reacts with two molecules of hydrochloric acid to produce one molecule of magnesium chloride and one molecule of hydrogen.
- 24 g of magnesium reacts with 73 g of hydrochloric acid to produce 95 g of magnesium chloride and 2 g of hydrogen.
- 24 g of magnesium reacts with 73 g of hydrochloric acid to produce 22.4 litres of hydrogen gas at S.T.P.
- It also proves the law of conservation of mass. According to the above equation, 97 gram of reactants are producing 97 gram of products.
(a) What are poly-atomic ions ? Give two examples.
(b) Name and state the fundamental law that every equation must fulfill.
Answer
(a) Polyatomic ions are ions composed of two or more atoms that are covalently bonded together and carry a net electrical charge. Examples — Nitrate ion (NO3-) and Sulphate ion (SO42-).
(b) Every equation must fulfill the "Law of Conservation of Matter". It states that matter is neither created nor destroyed in the course of a chemical reaction. Thus, the total mass of the substances on either side of the equation must be the same.
Give the information conveyed by the chemical formula of a compound.
Answer
By looking at the chemical formula, we understand the ratio in which different atoms are united to form the molecule.
Write the significance of a molecular formula.
Answer
The molecular formula of a compound has quantitative significance. It represents:
- both the molecule and the molecular mass of the compound.
- the respective numbers of different atoms present in one molecule of the compound.
- the ratios of the respective masses of the elements present in the compound.
For example, the formula CO2 means that:
- the molecular formula of carbon dioxide is CO2
- each molecule contains one carbon atom joined by chemical bonds with two oxygen atoms;
- the molecular mass of carbon dioxide is 44, given that atomic mass of carbon is 12 and that of oxygen is 16.
What do you understand by radicals : What are basic and acidic radicals ? Explain with examples.
Answer
A radical is an atom or a group of atoms of the same or of different elements that behaves as a single unit with a positive or a negative charge.
An Acid radical is the radical that remains after an acidic molecule loses a hydrogen ion (H+). Acid radicals typically have a negative charge. They are also called electronegative radicals or anions.
A Basic radical is the radical that remains after a base molecule loses a hydroxyl ion (OH-). Basic radicals typically have a positive charge. They are also called electropositive radicals or cations.
For example, in the compound ammonium carbonate (NH4)2CO3, ammonium (NH4+) is a basic radical with combining power 1 and carbonate (CO32-) is an acidic radical with combining power 2.
Give four examples of compounds with variable valency.
Answer
| Metal | Valency | Name of compound formed | Formula |
|---|---|---|---|
| Iron | 2 3 | Ferrous - [Iron (II)] oxide Ferric - [Iron (III)] oxide | FeO Fe2O3 |
| Copper | 1 2 | Cuprous - [Copper (I)] oxide Cupric- [Copper (II)] oxide | Cu2O CuO |
| Mercury | 1 2 | Mercurous - [Mercury (I)] oxide Mercuric - [Mercury(II)] oxide | Hg2O HgO |
| Lead | 2 4 | Plumbous - [Lead (II)] oxide Plumbic - [Lead (IV)] oxide | PbO PbO2 |
(a) Account for the existence of radicals in the atmosphere.
(b) How are they produced in the atmosphere?
(c) What is the role of radicals in the atmosphere?
Answer
(a) The major radicals present in the atmosphere are:
(i) Hydroxyl radical (OH●): It is also called as detergent of the atmosphere because it helps in removing air pollutants like methane, carbon monoxide and other harmful gases.
(ii) Nitrate radical (NO3●): It converts harmful gases into forms that can be washed out by rain.
(iii) Chlorine radical (Cl●): It is released from chlorofluorocarbons and reacts to destroy ozone layer.
(b) Atmospheric radicals are formed when stable molecules absorb energy, mainly from sunlight, and their chemical bonds break. This process produces highly reactive species with unpaired electron.
(c) Atmospheric radicals play a crucial role in both cleaning the atmosphere and influencing important environmental processes.
Free radicals are necessary for some biological functions, but excessive radicals cause significant damage. State the ways in which they can cause damage.
Answer
Excessive radicals cause following damages :
- Excess free radicals can damage cell membranes.
- They can harm proteins and enzymes.
- They can affect DNA and genetic material.
- They speed up ageing processes.
It is necessary to maintain a healthy balance of atmospheric radicals. Evaluate this statement.
Answer
A healthy balance of atmospheric radicals is necessary because some radicals help in cleaning the atmosphere, while others can harm it.
Hydroxyl radicals (OH•) and nitrate radicals (NO₃•) remove pollutants such as methane, carbon monoxide and other harmful gases from the air. However, excess chlorine radicals (Cl•) released from chlorofluorocarbons can destroy the ozone layer.
Hence, atmospheric radicals must be maintained in balance to keep the atmosphere clean without causing environmental damage.
Elements X, Y and Z have 3, 7 and 6 electrons in their valence shells respectively. Write the formula of the compound formed between :
(a) X and Y
(b) X and Z
Answer
(a) Valency of X is +3 and Y is -1. Formula of the compound formed between X and Y is :
∴ Chemical formula of the compound formed between X and Y is XY3
(b) Valency of X is +3 and Z is -2. Formula of the compound formed between X and Z is :
∴ Chemical formula of the compound formed between X and Z is X2Z3
The following figure represents the structural formula of a chemical compound.

Answer the questions given below:
(a) How many Carbon and Hydrogen atoms are present in the compound.
(b) Write the molecular and empirical formulae of the compound.
(c) Calculate the percentage composition of all the elements present in the compound. [At. wt. of C = 12, H = 1]
Answer
(a) 6 Carbon and 12 Hydrogen atoms are present.
(b) Its molecular formula is C6H12.
As the simplest ration between C and H is 1:2, hence the empirical formula is CH2
(c) Relative molecular mass of C6H12
= (6 x 12) + (1 x 12)
= 72 + 12
= 84 g
84 g of C6H12 contains 72 g of Carbon
∴ 100 g of C6H12 contains g of Carbon
= = 85.7 g of Carbon.
84 g of C6H12 contains 12 g of Hydrogen
∴ 100 g of C6H12 contains g of Hydrogen
= = 14.3 of Hydrogen.
∴ In C6H12, C = 85.7% and H = 14.3%
Calculate the molecular mass of the following :
(i) Na2SO4.10H2O
(ii) (NH4)2CO3
(iii) (NH2)2CO
(iv) Mg3N2
Given atomic mass of Na = 23, H = 1, O = 16, C = 12, N = 14, Mg = 24, S = 32
Answer
(i) Molecular mass of Na2SO4.10H2O
= 2 x 23 + 32 + 4 x 16 + 10(2 x 1 + 16)
= 46 + 32 + 64 + 180
= 322 amu
(ii) Molecular mass of (NH4)2CO3
= 2(14 + 4 x 1) + 12 + 3 x 16
= 36 + 12 + 48
= 96 amu
(iii) Molecular mass of (NH2)2CO
= 2(14 + 2 x 1) + 12 + 16
= 32 + 12 + 16
= 60 amu
(iv) Molecular mass of Mg3N2
= 3 x 24 + 2 x 14
= 72 + 28
= 100 amu
Calculate the relative molecular masses of
(a) CHCl3
(b) (NH4)2Cr2O7
(c) CuSO4.5H2O
(d) (NH4)2SO4
(e) CH3COONa
(f) Potassium chlorate, KClO3
(g) Ammonium chloroplatinate, (NH4)2PtCl6
[At. mass : C = 12, H = 1, O = 16, Cl = 35.5, N = 14, Cu = 63.5, S = 32, Na = 23, K = 39, Pt = 195, Ca = 40, P = 31, Mg = 24, Cr = 52]
Answer
(a) The relative molecular mass of CHCl3
= 12 + 1 + 3 x 35.5
= 12 + 1 + 106.5
= 119.5 amu
(b) The relative molecular mass of (NH4)2Cr2O7
= 2(14 + 4 x 1) + 2 x 52 + 7 x 16
= 36 + 104 + 112
= 252 amu
(c) The relative molecular mass of CuSO4.5H2O
= 63.5 + 32 + 4 x 16 + 5(2 x 1 + 16)
= 63.5 + 32 + 64 + 90
= 249.5 amu
(d) The relative molecular mass of (NH4)2SO4
= 2(14 + 4 x 1) + 32 + 4 x 16
= 36 + 32 + 64
= 132 amu
(e) The relative molecular mass of CH3COONa
= 12 + 3 + 12 + 16 + 16 + 23
= 82 amu
(f) The relative molecular mass of KClO3
= 39 + 35.5 + 3 x 16
= 39 + 35.5 + 48
= 122.5 amu
(g) The relative molecular mass of (NH4)2PtCl6
= 2(14 + 4 x 1) + 195 + 6 x 35.5
= 36 + 195 + 213
= 444 amu
Find the percentage mass of water in Epsom salt, MgSO4.7H2O.
Answer
Relative molecular mass of MgSO4.7H2O
= 24 + 32 + 4 x 16 + 7(2 x 1 + 16)
= 24 + 32 + 64 + 126
= 246 amu
246 g of Epsom salt contains 126 g of water of crystallisation
∴ 100 g of Epsom salt contains
= = 51.22 g of water of crystallisation.
∴ % mass of water in Epsom salt, MgSO4.7H2O = 51.22%
Calculate the percentage of phosphorus in:
(a) Calcium hydrogen phosphate, Ca(H2PO4)2
(b) Calcium phosphate, Ca3(PO4)2
Answer
(a) Relative molecular mass of Ca(H2PO4)2
= 40 + 2(2 x 1 + 31 + 4 x 16)
= 40 + 2(2 + 31 + 64)
= 40 + 194
= 234 amu
Wt. of P in Ca(H2PO4)2 = 2 x 31 = 62 g
% of P =
= x 100 = 26.5%
∴ Phosphorus in Calcium hydrogen phosphate is 26.5%
(b) Relative molecular mass of Calcium phosphate, Ca3(PO4)2
= 3 x 40 + 2(31 + 4 x 16)
= 120 + 2(31 + 64)
= 120 + 190
= 310 amu
Wt. of P in Ca3(PO4)2 = 2 x 31 = 62 g
% of P =
= x 100 = 20%
∴ Phosphorus in Calcium phosphate is 20%
Calculate the percentage composition of each element in Potassium chlorate, KClO3.
Answer
Relative molecular mass of KClO3
= 39 + 35.5 + 3 x 16
= 39 + 35.5 + 48
= 122.5 amu
122.5 g of KClO3 contains 39 g of Potassium
∴ 100 g of KClO3 contains g of Potassium
= = 31.83 g of Potassium
122.5 g of KClO3 contains 35.5 g of Chlorine
∴ 100 g of KClO3 contains g of Chlorine
= = 28.98 g of Chlorine
122.5 g of KClO3 contains 48 g of Oxygen
∴ 100 g of KClO3 contains g of Oxygen
= = 39.18 g of Oxygen
∴ In KClO3 : K = 31.83%, Cl = 28.98% and O = 39.18%
Urea is a very important nitrogenous fertilizer. Its formula is CON2H4. Calculate the percentage of carbon in urea.
(C= 12, O = 16, N = 14 and H = 1)
Answer
Relative molecular mass of CON2H4
= 12 + 16 + 2 x 14 + 4 x 1
= 12 + 16 + 28 + 4
= 60 amu
Wt. of C in CON2H4 = 12 g
% of C =
= x 100 = 20%
∴ Carbon in Urea is 20%
Calculate the percentage of iron in Ferric oxide Fe2O3.
Answer
Relative molecular mass of Ferric oxide (Fe2O3)
= 56 x 2 + 16 x 3
= 112 + 48
= 160 amu
Since 160 g of Ferric oxide (Fe2O3 ) contains 112 g of iron
∴ 100 g of contains Ferric oxide (Fe2O3 ) contains x 100 = 70%
∴ Percentage of Iron in Ferric oxide (Fe2O3 ) is 70%
Calculate amount of Nitrogen in one bag (50 kg) of Urea.
Answer
Molecular formula of Urea is CON2H4
Relative molecular mass of CON2H4
= 12 + 16 + 2 x 14 + 4 x 1
= 12 + 16 + 28 + 4
= 60 amu
Wt. of N in CON2H4 = 28 g
% of N =
= x 100 = 46.67%
Wt. of N in 50 kg CON2H4
= x 50 = 23.33 kg
∴ Amount of Nitrogen in one bag (50 kg) of Urea is 23.33 kg.
Calculate percentage of platinum in [(NH4)2 PtCl6]. Ammonium chloride platinate in nearest whole number.
Answer
The relative molecular mass of (NH4)2 PtCl6
= 2(14 + 4 x 1) + 195 + 6 x 35.5
= 36 + 195 + 213
= 444 amu
Wt. of Pt in [(NH4)2 PtCl6] = 195 g
∴ Percentage of platinum in (NH4)2PtCl6 is
= x 100
= 43.91 %
∴ Rounding to the nearest whole number, the percentage of platinum in ammonium chloride platinate (NH4)2 PtCl6 is 44%.
Calculate percentage composition of the elements in calcium phosphate Ca3(PO4)2.
Answer
Relative molecular mass of Calcium phosphate, Ca3(PO4)2
= 3 x 40 + 2(31 + 4 x 16)
= 120 + 2(31 + 64)
= 120 + 190
= 310 amu
310 g of Ca3(PO4)2 contains 120 g of Calcium
∴ 100 g of Ca3(PO4)2 contains g of Calcium
= = 38.70 % of Calcium.
310 g of Ca3(PO4)2 contains 62 g of Phosphorus
∴ 100 g of Ca3(PO4)2 contains g of Phosphorus
= = 20 % of Phosphorus.
310 g of Ca3(PO4)2 contains 128 g of Oxygen.
∴ 100 g of Ca3(PO4)2 contains g of Oxygen.
= = 41.3 % of Oxygen.
∴ In Ca3(PO4)2 : Ca = 38.70%, P = 20% and O = 41.3%
Assertion (A): The atomic mass of sodium is 23 amu.
Reason (R): An atom of sodium is 23 times heavier than an atom of carbon with mass 12 amu.
- Both A and R are true and R is the correct explanation of A.
- Both A and R are true but R is not the correct explanation of A.
- A is true but R is false.
- A is false but R is true.
Answer
A is true but R is false.
Explanation — The atomic mass of sodium is 23 amu. Hence, the assertion (A) is true.
An atom of sodium is 23 times heavier than one-twelfth the mass of a carbon-12 atom, not 23 times heavier than an atom of carbon with mass 12 amu. Hence, reason (R) is false.
Therefore, A is true but R is false is the correct option.
Assertion (A): An atom is the smallest part of matter which can take part in a chemical reaction.
Reason (R): Atoms of every element can exist independently.
- Both A and R are true and R is the correct explanation of A.
- Both A and R are true but R is not the correct explanation of A.
- A is true but R is false.
- A is false but R is true.
Answer
A is true but R is false.
Explanation — An atom is the smallest particle of an element and can take part in a chemical reaction. Hence, the assertion (A) is true.
Atoms may or may not exist independently.
For example:
Noble gas atoms like He, Ne can exist freely.
But atoms like H, O, and N do not exist alone under normal conditions; they exist as diatomic molecules (H2, O2, N2). Hence, reason (R) is false.
Therefore, A is true but R is false is the correct option.
Assertion (A): All equations need to be balanced.
Reason (R): An unbalanced equation would imply that atoms have been created or destroyed.
- Both A and R are true and R is the correct explanation of A.
- Both A and R are true but R is not the correct explanation of A.
- A is true but R is false.
- A is false but R is true.
Answer
Both A and R are true and R is the correct explanation of A.
Explanation — An equation must be balanced in order to comply with the "Law of Conservation of Matter", which states that matter is neither created nor destroyed in the course of a chemical reaction. Hence, the assertion (A) is true.
An unbalanced equation would imply that atoms have been created or destroyed. So, R us true.
Hence, both assertion (A) and reason (R) are true and reason (R) is the correct explanation of assertion (A).
Assertion (A): PCl3, is known as phosphorus trichloride while AlCl3, is aluminium chloride and not aluminium trichloride.
Reason (R) : Phosphorus shows variable valency. Aluminium does not show variable valency
- Both A and R are true and R is the correct explanation of A.
- Both A and R are true but R is not the correct explanation of A.
- A is true but R is false.
- A is false but R is true.
Answer
Both A and R are true and R is the correct explanation of A.
Explanation — A is true as phosphorus can show variable valency (like 3 and 5), so we name its compounds with prefixes like phosphorus trichloride (PCl3).
R is true as aluminium, however, shows a fixed valency of 3, so AlCl3 is simply called aluminium chloride without needing a prefix.
Hence, both assertion (A) and reason (R) are true, and reason (R) correctly explains assertion (A).
Assertion (A): In a chemical reaction, the total mass of the products remains the same as that of the reactants.
Reason (R): A chemical reaction involves a simple exchange of partners and no new species are formed.
- Both A and R are true and R is the correct explanation of A.
- Both A and R are true but R is not the correct explanation of A.
- A is true but R is false.
- A is false but R is true.
Answer
Both A and R are true but R is not the correct explanation of A.
Explanation — In a chemical reaction, the total mass of the substances on either side of the equation is the same, this is referred to be as law of conservation of mass. Hence, the assertion (A) is true.
In a chemical reaction the products are formed by the rearrangement of atoms or elements in the reactants. So, R is true.
Hence, Both A and R are true but R is not the correct explanation of A is the correct option.
Assertion (A): Magnesium phosphate is written as Mg3(PO4)2.
Reason (R): The valencies of magnesium and phosphate are three and two respectively.
- Both A and R are true and R is the correct explanation of A.
- Both A and R are true but R is not the correct explanation of A.
- A is true but R is false.
- A is false but R is true.
Answer
A is true but R is false.
Explanation — The molecular formula of Magnesium phosphate is Mg3(PO4)2. Hence, the given assertion (A) is true.
Given the formula Mg3(PO4)2 :
∴ Valency of magnesium (Mg) is 2 and Phosphate is 3. Hence, the reason (R) is false.
Therefore, A is true but R is false is the correct option.
Assertion (A): A chemical equation is written to represent molecules.
Reason (R): This is necessary for the law of conservation of mass
- Both A and R are true and R is the correct explanation of A.
- Both A and R are true but R is not the correct explanation of A.
- A is true but R is false.
- A is false but R is true.
Answer
Both A and R are true but R is not the correct explanation of A.
Explanation — A chemical equation represents molecules and it is necessary for the chemical equation to follow the law of conservation of mass. Hence, both assertion (A) and reason (R) are true.
However, balancing the equation is what ensures the law of conservation of mass. Therefore, reason (R) is not the correct explanation of assertion (A).
Therefore, Both A and R are true but R is not the correct explanation of A is not the correct option.
Assertion (A): Chemical combination always takes place between two elements.
Reason (R): A single substance is formed in a chemical combination.
- Both A and R are true and R is the correct explanation of A.
- Both A and R are true but R is not the correct explanation of A.
- A is true but R is false.
- A is false but R is true.
Answer
A is false but R is true.
Explanation — Chemical combination may take place between two or more elements. Hence, the assertion (A) is false.
A compound is a substance formed by the chemical combination of two or more elements in a fixed proportion. So, a single substance is formed in a chemical combination. Hence, the reason (R) is true.
Thereofore, A is false but R is true is the correct option.
Assertion (A): Hydroxyl radicals (OH•) are considered the "detergents" of the atmosphere.
Reason (R): Hydroxyl radicals are highly reactive and oxidise many pollutants, breaking them down into less harmful substances.
- Both A and R are true and R is the correct explanation of A.
- Both A and R are true but R is not the correct explanation of A.
- A is true but R is false.
- A is false but R is true.
Answer
Both A and R are true and R is the correct explanation of A.
Explanation — A is true as hydroxyl radicals (OH•) are called the “detergents” of the atmosphere. This is true because they help clean the air.
R is true as (OH•) radicals are highly reactive and oxidise many pollutants like methane and carbon monoxide, breaking them down into less harmful substances that can be removed from the atmosphere.
Therefore, Both A and R are true and R is the correct explanation of A is the correct option.
Assertion (A): The concentration of free radicals in the atmosphere is high during the night.
Reason (R): Free radicals are produced by the photolysis of molecules by solar radiation.
- Both A and R are true and R is the correct explanation of A.
- Both A and R are true but R is not the correct explanation of A.
- A is true but R is false.
- A is false but R is true.
Answer
A is false but R is true.
Explanation — Free radicals in the atmosphere are mainly formed when stable molecules absorb energy from sunlight and undergo photolysis. Hence, the R is true.
However, the statement that the concentration of free radicals is high during the night is not generally correct because photolysis depends on solar radiation, which is absent at night. Hence, the A is false.
Therefore, A is false but R is true is the correct option.
Atoms being extremely small, cannot be seen or weighed directly. But indirect methods of physics have enabled us to know the absolute mass of nearly all kinds of atoms. The mass of a hydrogen atom is found to be 1.66 × 10-24 g while that of a carbon atom is 1.9926 × 10-23 g. As these masses are too small, it is not convenient to use kilograms or grams as unit. It has, therefore, been considered appropriate to use the mass of some standard atom as a unit and then relate masses of other atoms to it.
(a) What do you understand by the statement "R.A.M. of silver is 108" ?
(b) Which element is considered as the standard for atomic masses ?
(c) What is the difference between R.A.M. and R.M.M.? Give an example to explain.
(d) What is the significance of amu or μ ?
(e) How is 1 amu related to grams ?
(f) Is the atomic mass expressed in amu the actual mass of an atom of that element ?
(g) Why is amu preferred instead of grams or kilogram for calculating the atomic mass of an atom ?
Answer
(a) “R.A.M. of silver is 108” means that one atom of silver is 108 times as heavy as 1⁄12th the mass of one carbon-12 atom.
(b) The element considered as the standard for atomic masses is carbon-12.
(c) R.A.M. means Relative Atomic Mass. It tells us how many times one atom of an element is heavier than th the mass of one carbon-12 atom.
Example:
R.A.M. of oxygen = 16.
This means one atom of oxygen is 16 times heavier than th the mass of one carbon-12 atom.
R.M.M. means Relative Molecular Mass. It tells us how many times one molecule of a substance is heavier than th the mass of one carbon-12 atom.
Example:
R.M.M. of water, H2O:
R.M.M. of H2O = 2 × R.A.M. of H + 1 × R.A.M. of O
= 2 × 1 + 16 = 18
So, the R.M.M. of water is 18. This means one molecule of water is 18 times heavier than th the mass of one carbon-12 atom.
Thus, R.A.M. is used for atoms of elements, while R.M.M. is used for molecules of elements or compounds.
(d) The significance of amu is that it provides a convenient unit for expressing the extremely small masses of atoms and molecules. It simplifies calculations by avoiding extremely small values in grams.
(e) 1 amu = 1.66 × 10-24 g
(f) No, the atomic mass expressed in amu is not the actual mass of an atom in grams. It is a relative mass expressed by comparing the mass of the atom with th the mass of a carbon-12 atom.
(g) The amu is preferred because the actual masses of atoms in grams or kilograms are extremely small and inconvenient to use. The amu gives a simple, practical and manageable scale for comparing atomic masses.
An element X forms a chloride with the formula XCl3, and an oxide with the formula X2O3. This oxide dissolves in water to form a solution that turns blue litmus red.
(a) Identify the element.
(b) Is X a metal or a non-metal ? Justify your answer.
(c) Write the chemical formula for the sulphate of element X.
Answer
(a) The element is phosphorus.
Reason — In XCl3, chlorine has valency 1. Since one atom of X combines with three chlorine atoms, the valency of X is 3.
But its oxide turns blue litmus red which means it's a non-metal. Phosphorus is a non-metal with valency 3. Formula for phorphorus oxide is P2O3 . Boron and aluminium also have valency 3 but boron is a metalloid and aluminium is a metal.
(b) X is a non-metal because its oxide forms an acidic solution in water (turns blue litmus red), which is a characteristic property of non-metals.
(c) The sulphate of phosphorus is P2(SO4)3.
Note — Although the formula P2(SO4)3 can be written using valency rules, this compound does not actually exist in reality. This is because sulphates are formed by metals (which give cations), whereas phosphorus is a non-metal and does not form P3+ cations under normal conditions. Hence, P2(SO4)3 is only a theoretical formula based on valency.
A compound is found to have a mass ratio of element A to element B as 3 : 8. If the formula of the compound is AB, what is the relationship between the atomic masses of A and B.
Answer
Atomic mass of A = MA
Atomic mass of B = MB
Since the formula of the compound is AB, one atom of A combines with one atom of B.
∴ MA : MB = 3 : 8
or,
On cross-multiplication,
8MA = 3MB
Thus, the relationship between the atomic masses of A and B is
8MA = 3MB